A Rare Case of LMCA Stenosis Treated by Percutaneous Coronary Intervention (PCI) in Setting of Patent Ductus Arteriosus with Dilated Pulmonary Trunk
Sushant Wattal1, Abdul UK Razak2, Sudhakar Rao Mugula3, Ranjan Kediyoor Shetty4, Padmakumar Ramachandran5
1 Resident, Department of Cardiology, KMC, Manipal University, Manipal, Karnataka, India.
2 Assistant Professor, Department of Cardiology, KMC, Manipal University, Manipal, Karnataka, India.
3 Resident, Department of Cardiology, KMC, Manipal University, Manipal, Karnataka, India.
4 Professor, Department of Cardiology, KMC, Manipal University, Manipal, Karnataka, India.
5 Professor, Department of Cardiology, KMC, Manipal University, Manipal, Karnataka, India.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Dr. Sudhakar Rao Mugula, Resident, Department of Cardiology, Manipal-576104, Karnataka, India.
E-mail: msudhakar88@gmail.com
Extrinsic compression of the Left Main Coronary Artery (LMCA) by a dilated pulmonary artery is a rare but a treatable cause of angina in patients with Pulmonary Artery Hypertension (PAH). In majority of these patients, angina is attributed to right ventricular or left ventricular demand ischemia and in the absence of cardiovascular risk factors for atherosclerosis, they rarely undergo a coronary angiogram. So this rare diagnosis may be missed in such patients and optimal management delayed. In this case report we describe a patient of PAH secondary to Patent Ductus Arteriosus (PDA), presenting with heart failure and complaining of persistent angina, due to extrinsic compression of the ostial LMCA by the dilated pulmonary artery, treated successfully by Percutaneous Coronary Intervention (PCI). This case reminds us that in all patients of pulmonary hypertension presenting with angina and left ventricular dysfunction, this rare but treatable cause should be identified by coronary angiography and treated with PCI.
Angina, Left ventricular dysfunction, Ostial stenosis
Case Report
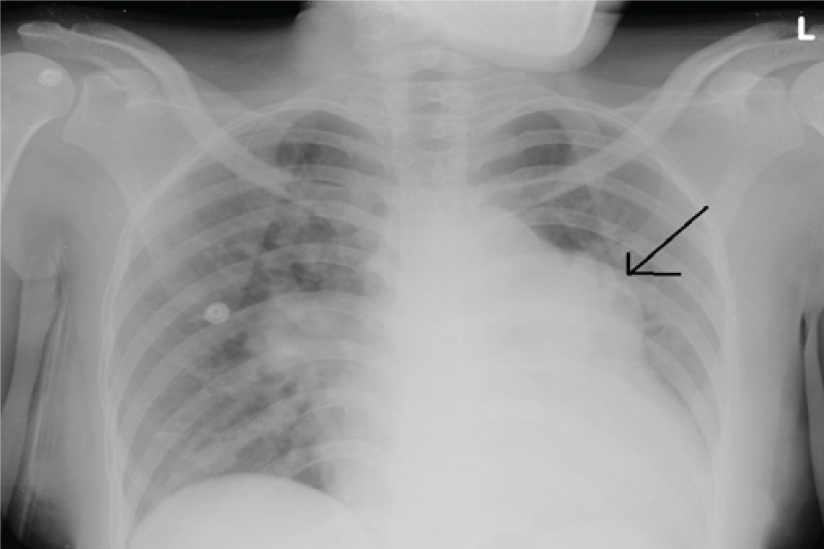
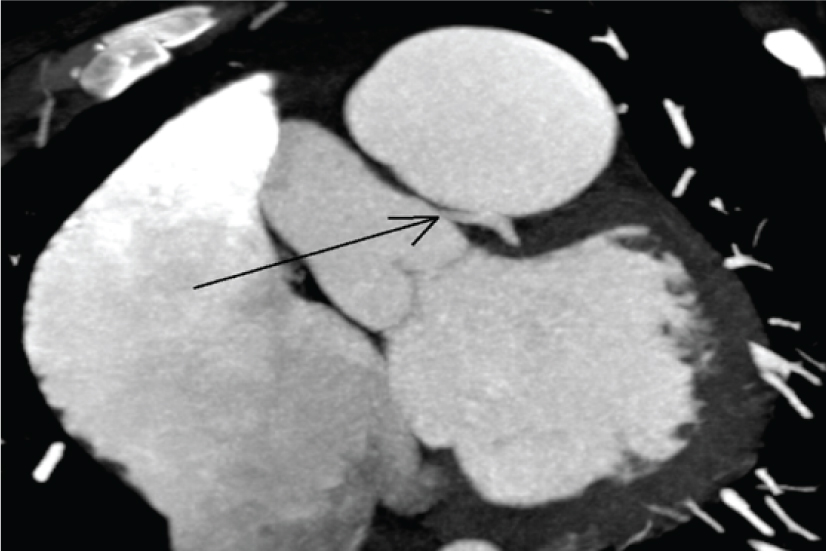
A 23-year-old female, 36 weeks primigravida presented with complaints of breathlessness since 1 week. There was no history of any bluish discoluration of skin or mucous membrane in the past. On examination, there was no cyanosis or clubbing but she had raised Jugular Venous Pressure (JVP) and bilateral basal crepitations were audible in the chest. Cardiovascular examination showed a loud pulmonary component of second heart sound and a systolic murmur was heard in the left upper sternal border. Chest X-ray showed cardiomegaly with enlarged pulmonary conus and pulmonary oedema [Table/Fig-1]. Electrocardiogram showed biventricular hypertrophy. A two dimensional Echocardiogram showed 8 mm PDA with Bi-directional but predominantly left to right shunt with severe tricuspid regurgitation and severe PAH. Right atrium, ventricle and pulmonary artery were dilated and bi-ventricular dysfunction was present. She was managed medically with intravenous diuretics and her heart failure improved. Ten days later she had similar symptoms of increasing breathlessness and had to undergo emergency LSCS. She delivered a healthy male neonate. Two months later patient came for follow up for further management of PDA. In addition to shortness of breath on exertion (NYHA class III). She had no other associated cardiovascular risk factors. CT pulmonary angiogram showed main pulmonary artery (44.9 mm), right pulmonary (30.2 mm) and left pulmonary (26.7 mm) arteries were grossly dilated in calibre. A PDA of length 5.5 mm and width of 8.8 mm between descending aorta and main pulmonary trunk distal to the origin of subclavian artery was seen. CT Coronary angiogram showed external LMCA compression by the dilated pulmonary artery [Table/Fig-2]. Cath study was done which showed a Qp/Qs of 2.6 with normal pulmonary artery pressures and was suggestive of hyperkinetic PAH. In view of the large size of PDA, coiling was not considered. However, balloon occlusion of PDA was tried but there was a rapid rise in the left ventricular end diastolic pressure resulting in the development of pulmonary oedema. In view of the same she was continued on medical management.
Chest X-Ray PA view showing cardiomegaly with enlarged pulmonary conus and pulmonary oedema.
CT Coronary angiogram showing external left main coronary artery compression by the dilated pulmonary artery.
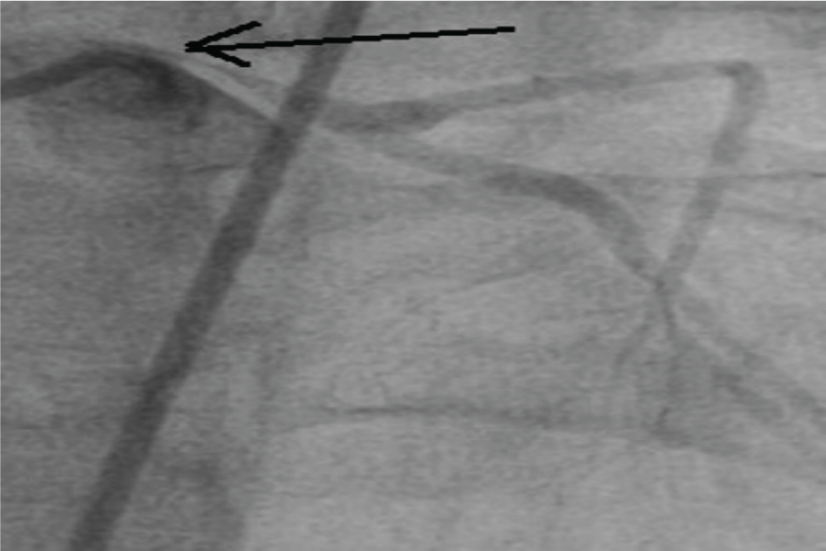
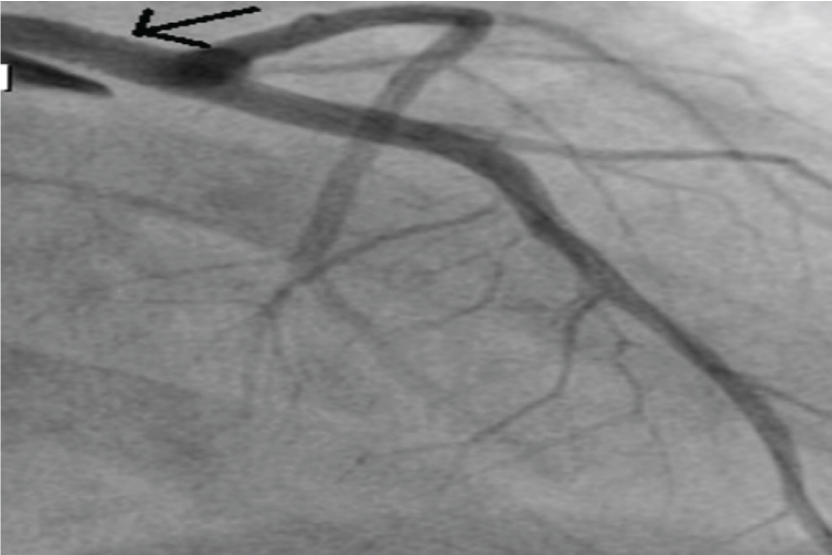
Another four months later she presented with rest angina. Coronary angiogram was done which showed 90% stenosis of the left main coronary artery ostium in the form of a “pencil point” that progressively recovered its diameter [Table/Fig-3]. The rest of the coronary tree was normal with no evidence of arteriosclerosis. In view of persistent angina and LV dysfunction, external compression of ostial LMCA from the dilated Main Pulmonary Artery was considered as the cause and it was successfully stented with a 4*18 mm ever olimus eluting stent at 14 atm. Post-percutaneous coronary intervention results were good and no residual stenosis of the LMCA was noted [Table/Fig-4]. Patient was started on dual antiplatelet therapy and continued on decongestive measures. Patient got relieved from angina subsequently and LV function also improved following successful angioplasty of the LMCA. This case illustrates a rare example of persistent angina in severe PAH in PDA due to extrinsic compression of LMCA by a dilated pulmonary artery successfully relieved with angioplasty.
Coronary angiogram in AP cranial view showing 90% stenosis of the left main coronary artery ostiin the form of a “pencil point”.
Coronary angiogram in AP cranial view after percutaneous intervention showing well expanded stent in left main coronary artery with TIMI 3 flow.
Discussion
Chest pain is a common symptom in patients with PAH, seen in approximately 40% of patients [1]. Right Ventricular (RV) hypertrophy leading to intra-myocardial compression of the arterioles or a decrease in the coronary perfusion gradient due to high right atrial pressure causing demand ischemia of the RV is the most commonly attributable explanation. Another explanation given for these pains is the acute pulmonary artery dilatation occurring during transitory increases in pulmonary artery pressure [2].
Extrinsic compression of the Left Main Coronary artery due to a dilated main pulmonary artery is a very important, and moreover a potentially reversible cause of angina and left ventricular dysfunction in patients with pulmonary hypertension [3]. Therefore, early detection and management can lead to rapid symptomatic improvement, as seen in our case. Also, the malignant arrhythmia and left ventricular dysfunction caused by LMCA compression may lead to a higher incidence of sudden death in patients with pulmonary hypertension [4].
Left main compression syndrome in pulmonary hypertension has been reported in various case reports and case series [2,5-10]. Kajita LJ et al., reported series of 12 cases of LM extrinsic compression in patients with pulmonary artery dilatation of different aetiology. The majority was with the congenital heartdisease (ASD, VSD, Tetralogy of Fallot, PDA) [11].
In our case, it was PDA with secondary PAH. However, case reports about LMCA compression due to enlarged pulmonary artery in PAH secondary to PDA are very few, making this case a rare one [6,8,10].
The incidence of LMCA compression due to PA dilatation is not well known, but according to different case series it may range between 5% and 44% [12,13].
Unlike atherosclerotic coronary artery disease, which is more seen in elderly males, extrinsic LMCA compression affects younger people and occurs more commonly in women, as seen in this case [11]. Apart from younger age group, severe pulmonary trunk dilatation (>40 mm; normal: 25–30 mm), also predisposes to LMCA compression in PAH, as seen in this case where pulmonary artery diameter was 44 mm [13].
Optimal treatment for primary pulmonary hypertension in patients with extrinsic LMCA compression has not been clearly established. Rich S et al., reported 2 cases of patients with PAH and extrinsic LMCA compression treated by Percutaneous intra coronary stenting [5]. Similar to our case, the angiographic result was very good. Also, one of the patients had severe left ventricular systolic dysfunction, which normalized after the procedure. Therefore, as seen in this case myocardial ischemia may be the reason behind left ventricular systolic dysfunction when extrinsic compression of the LMCA is present.
Whether the functional and prognostic significance in these cases is similar to that for patients with occlusive arteriosclerosis involving the LMCA is unknown. However, as occurred with our patient, the angina may be relieved by stent revascularization, but no data is available on the long-term benefits of this technique.
Conclusion
Extrinsic compression of the LMCA is an underdiagnosed and a potentially treatable cause of angina in patients with PAH with dilated pulmonary artery. So a coronary angiogram should always be considered in patients with pulmonary hypertension having symptoms of exertion angina or left ventricular systolic dysfunction to identify this rare entity. Percutaneous coronary stenting of the externally compressed LMCA can be carried out with good angiographic results and can help relieve angina and improve LV function. However, its impact on the long term prognosis still needs more research.
[1]. Albadri K, Jensen JM, Christiansen EH, Mellemkjær S, Nielsen-Kudsk JE, Left main coronary artery compression in pulmonary artery hypertensionPulm Circ 2015 5(4):734-36. [Google Scholar]
[2]. Gomez Varela GS, Montes Orbe PM, Alcibar Villa AJ, Egurbide MV, Sainy I, Barrentexea Benguria JI, Stenting in primary pulmonary hypertension withcompression of the left main coronary arteryRev Esp Cardiol 2004 57(7):695-98. [Google Scholar]
[3]. Lee MS, Oyama J, Bhatia R, Kim YH, Park SJ, Left main coronary artery compression from pulmonary artery enlargement due to pulmonary hypertension: a contemporary review and argument for percutaneous revascularizationCatheter Cardiovasc Interv 2010 76(4):543-50. [Google Scholar]
[4]. Lindsey JB, Brilakis ES, Banerjee S, Acute coronary syndrome due to extrinsiccompression of the left main coronary artery in a patient with severe pulmonary hypertension: successful treatment with percutaneous coronary interventionCardiovasc Revasc Med 2008 9(1):47-51. [Google Scholar]
[5]. Rich S, McLaughlin VV, O’Neill W, Stenting to reverse left ventricular ischemia due to left main coronary artery compression in primary pulmonary hypertensionChest 2001 120(4):1412-15. [Google Scholar]
[6]. Kothandam S, Maruthanayagam R, Gnanapragasam F, Krishnaswami M, Velayudhan B, Extrinsic compression of the left coronary ostium by the pulmonary trunkTex Heart Inst J 2010 37(1):95-98. [Google Scholar]
[7]. Dubois CL, Dymarkowski S, Cleemput JV, Compression of the left main pulmonary artery in a patient with the Eisenmenger syndromeEur Heart J 2007 28(16):1945 [Google Scholar]
[8]. Dodd JD, Maree A, Palacios I, de Moor MM, Mooyaart EA, Shapiro MD, Images in cardiovascular medicine. Left main coronary artery compression syndrome: evaluation with 64-slice cardiac multidetector computed tomographyCirculation 2007 115(1):e7-8. [Google Scholar]
[9]. Ginghina C, Popescu BA, Enache R, Ungureanu C, Deleanu D, Platon P, Pulmonary artery dilatation: an overlooked mechanism for angina pectorisJ Cardiovasc Med 2008 9(7):747-50. [Google Scholar]
[10]. Caldera AE, Cruz-Gonzalez I, Bezerra HG, Cury RC, Palacios IF, Cockrill BA, Endovascular therapy for left main compression syndrome. Case report and literature reviewChest 2009 135(6):1648-1650. [Google Scholar]
[11]. Kajita LJ, Martinez EE, Ambrose JA, Lemos PA, Esteves A, NogueiradaGama M, Extrinsic compression of the left main coronary artery by a dilated pulmonary artery: clinical, angiographic, and hemodynamic determinantsCatheter Cardiovasc Interventions 2001 52(1):49-54. [Google Scholar]
[12]. Mitsudo K, Fujino T, Matsunaga K, Doi O, Nishihara Y, Awa J, Coronary arteriographic findings in the patients with atrial septaldefect and pulmonary hypertension (ASD + PH)—compression of leftmain coronary artery by pulmonary trunk [in Japanese]Kokyu To Junkan 1989 37(6):649-55. [Google Scholar]
[13]. Mesquita SM, Castro CR, Ikari NM, Oliveira SA, Lopes AA, Likelihood of left main coronary artery compression based on pulmonary trunk diameterin patients with pulmonary hypertensionAm J Med 2004 116(6):369-74. [Google Scholar]