Langerhans Cell Sarcoma of Thyroid: A Rare Differential for a Thyroid Swelling
Suhas K Rajappa1, Dharma Ram2, Makhan L Lohiya3, Sunil Pasricha4, Ajay K Dewan5
1 Consultant, Department of Surgical Oncology, Rajiv Gandhi Cancer Institute and Research Center, New Delhi, India.
2 Assistant Professor, Department of Surgical Oncology, Dr SN Medical College, Jodhpur, Rajasthan, India.
3 Principle Specialist, Department of General Surgery, Dr SN Medical College, Jodhpur, Rajasthan, India.
4 Consultant, Department of Surgical Oncology, Rajiv Gandhi Cancer Institute and Research Center, New Delhi, India.
5 Chief and Head, Department of Surgical Oncology, Rajiv Gandhi Cancer Institute and Research Center, New Delhi, India.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Dr. Dharma Ram, C 151 A Kamla Nehru Nagar, Jodhpur-342009, Rajasthan, India.
E-mail: drdharmapoonia@gmail.com
Langerhans Cell Sarcoma (LCS) is a rare disease arising from antigen-presenting cells. The literature on LCS is largely limited to case series and only 67 such cases have been reported till date. We report a case of 27-year-old female presenting as a thyroid swelling. Computerized tomography (CT) scan showed diffuse enlargement of thyroid and patient underwent total thyroidectomy. Final diagnosis on histopathology and Immunohistochemistry (IHC) was langerhans cell sarcoma of the thyroid. Patient was advised adjuvant chemotherapy, however she defaulted and later presented to us with a disseminated disease. This is the 18th case of head and neck LCS to be reported and the first case of primary thyroid origin in the world literature. The diagnosis of LCS is by combination of histopathology and IHC and multi- modality treatment offers the best chance of cure.
Immunohistochemistry, Lymphadenopathy, Rare sarcomas
Case Report
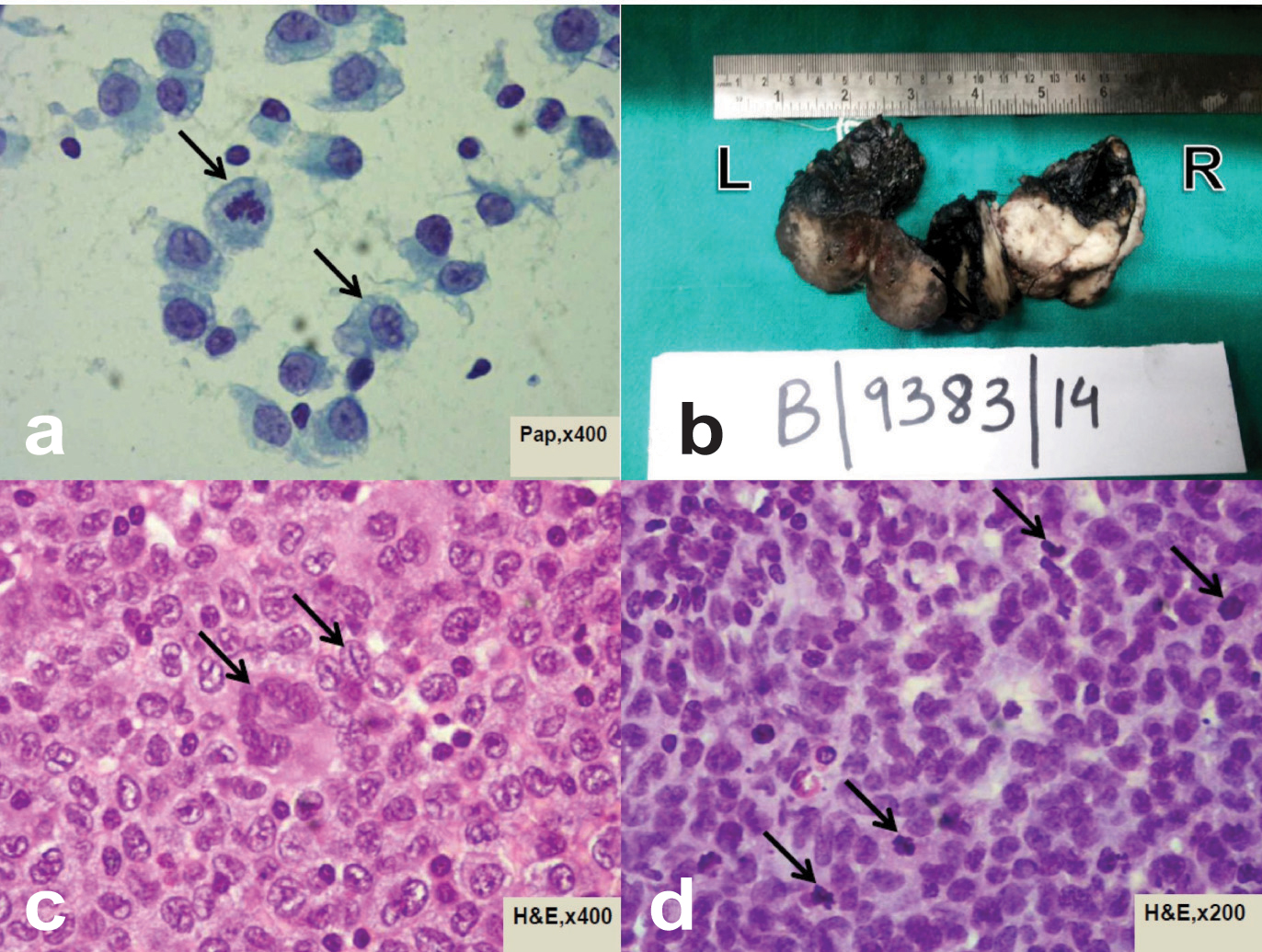
A 27-year-old female, known case of hypothyroidism presented with a complaint of swelling in the front region of the neck of one-year duration with recent onset pain of two months duration. On examination, there was a diffuse enlargement of the thyroid with no cervical lymphadenopathy. Thyroid function test was within normal limits. Fine needle aspiration showed cells predominantly dispersed, with focal aggregates of tumour cells comprising of large cells having irregular convoluted atypical nuclei with grooving and exhibiting significant mitosis [Table/Fig-1a]; overall findings were suggestive of a poorly differentiated malignancy (Bethesda category C-5). CT scan of the neck was done which showed diffuse enlargement of both the lobes of thyroid and isthmus (Right>Left) with some nodularity and homogeneous contrast enhancement. The patient underwent total thyroidectomy with central compartment clearance and post op recovery was uneventful. Final histopathology revealed a diffuse proliferation of medium to large neoplastic cells with vesicular nuclei, showing lobulated and convoluted outlines with brisk mitosis. Areas with low-grade nuclear features and prominent nuclear grooving were also evident. Tumour giant cells were also seen and there was associated lymphocytic thyroiditis [Table/Fig-1b-d].
Showing pathological finding of the tumour: a) FNAC showing large neoplastic cells showing convoluted nuclei with grooving and low-grade nuclear grade (Pap 40X); b) Gross pathology: Both the lobes are enlarged (Right>Left) and Cut surface of right and left lobe was grey white and tan brown respectively; c) tumour cells with vesicular and convoluted nuclei with few areas of grooving and tumour giant cells (H&E 40X); d) Different area showing hyperchromatic nuclei with brisk mitosis, suggestive of high-grade nuclear feature (H&E 20X).
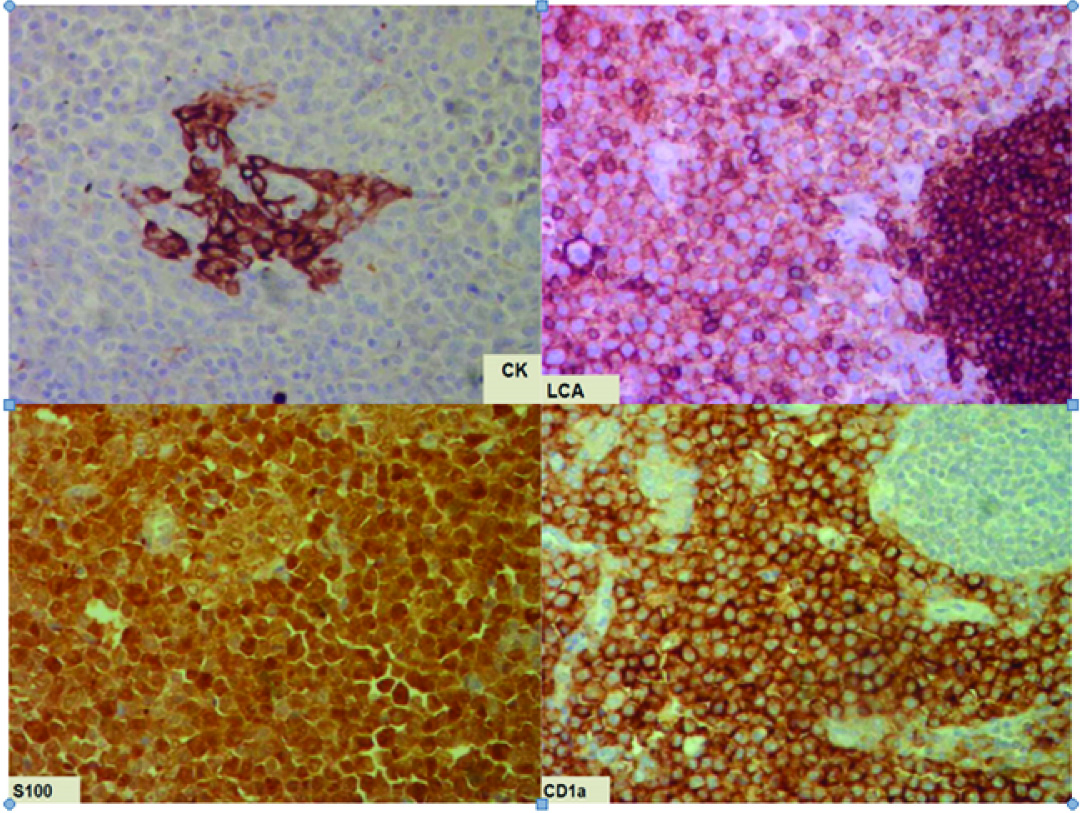
Histopathologically diagnosis was of a poorly differentiated malignancy. On evaluating the primary IHC panel the tumour cells were positive for LCA while negative for Creatine Kinase (CK), EMS, and Thyroid Transcription Factor (TTF) [Table/Fig-2], hence confirming the hematolymphoid malignancy. On further IHC the tumour cells were negative for B cell and T cell tumour markers and were also negative for Epithelial Membrane Antigen (EMA), CD 30, ALK, Terminal deoxynucleotidyl Transferase (TdT) hence ruling out almost all the categories of Non Hodgkins Lymphoma (NHL). However in view of significant nuclear grooving, IHC for S100 and CD 1a [Table/Fig-2] was performed and was found to be positive hence confirming the suspicion of LCS probably arising on the background of LCH. The Ki-67 index was 75%.
Immunohistochemistry panel of tumour showing positive for LCA (DAB, 20X), S100 (DAB, 20X) and CD1a (DAB, 20X); negative for CK (DAB, 20X), overall picture suggesting LCS.
Repeat clinical examination and metastatic workup were done which did not reveal any abnormalities. After a tumour board discussion patient was advised adjuvant chemotherapy but she was lost to follow up. The patient presented to us two years later with complaints of multiple ulcerative lesions in the oral cavity of one-month duration. Local examination revealed multiple verrucous lesions in bilateral buccal mucosa and an ulceroproliferative growth in hard palate. Biopsy was done which showed similar histopathological features and hence confirming the recurrence. PET-CT was done which showed Fludeoxyglucose (FDG) avid lesions in bilateral tonsils, bilateral cervical nodes and a lesion in left upper lobe of lung along with oral cavity lesions. The patient is currently on palliative chemotherapy.
Discussion
Langerhans cells are the antigen presenting cells of the body and the tumours arising from these cells can be benign or malignant. World Health Organization (WHO) classifies Langerhans cell tumours into Langerhans Cell Histiocytosis (LCH) and LCS; LCS presenting with overt features of malignancy like poor nuclear differentiation, brisk mitosis and high Ki-67 score [1]. The confirmation of Langerhans cell lineage requires IHC demonstration of markers like Langerin, CD1a or S-100 [2]. Historically, demonstration of Birbeck granules on electron microscopy was used as a marker of Langerhans cell but now it is not considered essential for diagnosis as many a time these granules may be absent [3]. As a part of the innate defense system of the body Langerhans cells are widely distributed and hence LCS can originate from any site but lymph nodal disease is the most commonly reported along with skin lesions. A total of about 67 cases of LCS have been reported till date and 17 cases among them were in head and neck [4]. Head and neck LCS predominantly affects the skin, lymph nodes, and tonsil and to the best of our knowledge this is the first case report in the world literature describing LCS of primary thyroid origin.
Exact aetiology is largely unknown while immunosuppression and transdifferentiation have been listed as the probable cause by few authors [5,6]. In our case, presence of low-grade areas resembling LCH raises a possibility of divergent differentiation or dedifferentiation into LCS resulting due to genomic instability. No imaging features are characteristic of the entity but the role of imaging is to define resectability and extent of the disease. As there is no specific pattern of spread and metastasis to various organs has been demonstrated in earlier reports, we suggest evaluation with PET-CT to be the standard mode of staging.
Because of its rarity, the management is largely debatable with multimodality treatment being the most commonly used with a good response. Total thyroidectomy with central nodal clearance was done in our patient and was advised for adjuvant therapy after tumour board discussion but patient defaulted and came back with a disseminated disease, which probably signifies the requirement of multimodality therapy in managing this rare disease. Whenever feasible, resection with adequate margins along with either sentinel node biopsy or nodal dissection should be the treatment of choice in localised disease [7]. There is an isolated case report of treating localised LCS with radiation only [3] but lack of further studies should caution against using this modality alone. Adjuvant treatment includes either chemotherapy alone or concurrent chemoradiation and the benefit is only seen if administered in timely fashion [4]. Many chemotherapeutic regimens have been tried for locally advanced LCS and the most common in head and neck cohorts being CHOP (cyclophosphamide/pirarubicin/vincristine/prednisolone) while few authors slightly modified the regimen by adding etoposide. The other regimen which has been tried is gemcitabine/docetaxel. Howard JEF et al., in his systemic review of previous case reports showed four out of six patients with locoregional disease had only surgery or chemotherapy and among those three patients expired and one is alive with the disease [7]; the other two patients for whom the surgery and chemotherapy were used, one achieved complete remission and other is alive with the disease. Even though adjuvant chemoradiation has been tried with good results in non-head and neck LCS it is yet to be tested in a non-metastatic head and neck LCS. Management of disseminated disease is usually unsatisfactory and it is fatal in almost all the cases. Palliative chemotherapy, chemoradiation has been tried without much survival benefit. There are instances of complete cure with bone marrow transplant [8] in a disseminated disease but these are case reports of infants and whether these could be translated into adults needs to be seen.
Conclusion
Langerhans cell sarcoma is a rare tumour with poor prognosis and requires diagnostic adroitness. This may be considered a rare differential for atypical thyroid cases. Multispecialty discussion helps in deciding the optimum management of rare diseases like LCS where there is a paucity of literature particularly of the individual subsite. Multimodality treatment is the best chance for cure in non-metastatic disease but further research is needed to decide on the appropriate chemotherapeutic regimen, sequencing of the treatment modalities and management of patients with the disseminated disease.
[1]. Swerdlow S, Campo E, Harris N, Jaffe E.S, Pileri S.A, Stein H, WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues 2008 4th edIARC Press [Google Scholar]
[2]. Pileri SA, Grogan TM, Harris NL, Banks P, Campo E, Chan JK, Tumours of histiocytes and accessory dendritic cells: An immunohistochemical approach to classification from the International Lymphoma Study Group based on 61 casesHistopathology 2002 41:1-29. [Google Scholar]
[3]. Nakayama M, Takahashi K, Hori M, Okumura T, Saito M, Yamakawa M, Langerhans cell sarcoma of the cervical lymph node: a case report and literature reviewAuris Nasus Larynx 2010 37:750-53. [Google Scholar]
[4]. Howard JEF, Dwivedi RC, Masterson L, Jani P, Langerhans cell sarcoma: A systematic review Cancer Treatment Reviews 2015 41:320-31. [Google Scholar]
[5]. Yoshimi A, Kumano K, Motokura T, Takazawa Y, Oota S, Chiba S, ESHAP therapy effective in apatient with langerhans cell sarcomaInt J Hematol 2008 87:532-37. [Google Scholar]
[6]. Muslimani A, Chisti M, Blenc AM, Boxwala I, Micale MA, Jaiyesimi I, Langerhans/dendritic cell sarcoma arising from hairy cell leukemia: a rare phenomenonAnn. Hematol 2012 91:1485-87. [Google Scholar]
[7]. Howard JEF, Dwivedi RC, Masterson L, Jani P, Langerhans cell sarcoma of the head and neck critical reviews in oncology/hematology 2016 99:180-88. [Google Scholar]
[8]. Chung WD, Im SA, Chung NG, Park GS, Langerhans cell sarcoma in two young children: Imaging findings on initial presentation and recurrenceKorean J Radiol 2013 14:520-24. [Google Scholar]