Therefore, the goal of this study was to evaluate and compare the effectiveness of three remineralizing agents: NaF varnish, n-HAP and NSF on enamel of primary anterior teeth. The null hypothesis was that there is no difference in remineralizing effect of these materials.
Materials and Methods
Sample preparation: This experimental in vitro study was approved by Ethics Review Committee of the School of Dentistry, Shiraz University of Medical Sciences (IR.SUMS.REC.1396.595) and conducted at the School of Dentistry from March to April 2017. The study sample size was calculated according to previous experimental studies [6,26]. Primarily a total of 80 sound primary anterior teeth were collected. Primary canines were extracted due to orthodontic supervision purposes. Over retained primary incisors were also extracted in cases of successor permanent incisor delayed eruption. The exclusion criteria were teeth with decay, crack and any enamel defect. The samples were examined using stereomicroscope (Motic K®, Wetzlar, Germany) and a number of nine samples were excluded because of these reasons. The crowns and the roots were separated from 2 mm below the Cemento-Enamel Junction (CEJ). The crowns were first washed of debris and also disinfected with 0.1% thymol. They were mounted with epoxy resin so that their buccal surfaces were exposed and to obtain a flat surface, the teeth were polished with silicon carbide paper (600-, 800-, and 2400-grit) respectively, and washed in distilled water for 20 seconds. In order to enclose the surface to an exposed area of 2 × 2 mm, two layers of nail varnish were applied in the surrounding area [29].
Baseline microhardness test and group classification: As the next step, Surface Microhardness (SMH) of the samples was tested by Vickers micro hardness tester (SCTMC®, MHV 10002, China) under a 50 gram load for 10 seconds. The surface microhardness was assessed at five random points on the enamel. The first measurement was performed at the center of the sample and the other ones were at a distance of 300 μm from the first indentation and then the mean for SMH was calculated [26,30]. SMH tests were conducted by an operator. Samples with SMH between 262.40 and 389.30 were selected and samples with SMH below that (11 samples) were excluded.A total of 60 samples were randomized into four groups of 15 samples each based on the applied remineralizing agents as the following: (1) NaF varnish (Duraphat®, Colgate, Sao Paulo); (2) n-HAP repairing serum (PrevDent International BV®, Netherlands); (3) NSF (Sigma-Aldrich®, Munich, Germany) and (4) control-no treatment.
Solution preparation: Demineralization solution: This solution was prepared with 50 mM acetic acid (pH 4.5), 2.2 mM potassium dihydrogen phosphate (KH2PO4), 2.2 mM calcium nitrate (Ca(NO3)2) and 0.1 ppm NaF.
Remineralization solution: The remineralization solution used in pH-cycling contained 20 mMol l−1 HEPES(4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid), 130 mM potassium chloride (KCl),1.5 mM calcium chloride (CaCl2), 0.9 mM potassium dihydrogen phosphate (KH2PO4) and 1 mM sodium azide (NaN3). The pH was adjusted to 7.0 with potassium hydroxide (KOH).
Incipient caries lesion formation: Before application of the remineralizing agents, incipient caries were generated. A pH-cycling protocol was performed as said by Ten Cate et al., [31]. Each tooth sample was stored in 30 ml of demineralization solution for three days (72 hours) at 370C. Then, the specimens were washed with distilled water for 20 seconds. SMH testing was performed again after the development of artificial caries with the same micro hardness tester machine (SCTMC®, MHV 10002, China) and the same operator, as the previous protocol [10].
Experimental groups and pH cycling model: After the second microhardness test, the samples were treated according to each groups’ manufactures’ instructions.
Group 1 (5% NaF Varnish): The varnish (Duraphat®, Colgate, Sao Paolo) was applied with microbrush and specimens were stored in artificial saliva for 24 hours. After that, the varnish was eliminated from the surface with a scalpel blade without scratching the enamel surface [26].
Group 2 (n-HAP Serum): The n-HAP serum (PrevDent International BV®, Netherlands) was applied with its sponge for three minutes according to manufacturer’s instruction. After this, the paste was kept in contact with the enamel surface of the specimens for 20 minutes. Then the samples were washed for 20 seconds with de-ionized water.
Group 3 (NSF): The solution was prepared by dilution of NSF powder (Sigma-Aldrich®, Munich, Germany) in distilled water (Ag+ {376.5 μg/ml}). Solution was left in contact with the tooth surface for two minutes. For each specimen two drops of NSF solution were applied with a micro brush [2].
Group 4 (Control): no agent was applied.
The cariogenic challenge was accomplished with the de- and remineralizing solutions described before. Specimens were maintained in remineralizing solution for 21 hours and in the demineralizing solution for three hours.
This cycle was repeated for 10 days [32]. The re- and demineralizing solutions were changed every third day. Before each immersion in demineralization solution, the different pastes were applied according to each group. The n-HAP serum was applied once daily for 10 days while the varnish and NSF solution were applied only once to mimic their single applications in clinical settings [2,26,32].
The post treatment SMH tests were conducted by the same operator blindly and with the same conditions (load, time and the machine) applied for baseline and post lesion measurements.
The surface microhardness recovery percentage was assessed as follows: % SMHR equals 100 {(post treatment-post lesion)/(baseline-post lesion)} [17].
Atomic force microscopy: After micro hardness testing, a total of 12 random specimens of all groups (three random samples of each group) were tested using atomic force microscope (NanoWizard® II; JPK Instrument AG, Berlin, Germany). The block surface morphology was analysed in “contact mode.”
Imaging was obtained by an AFM nonconductive silicon nitride cantilever (Acta-Probe, APPNano, CA) with a spring constant (13 N/m). The rate of scanning was 1 Hz and a piezoelectric scanner was used. Images of 15 x 15 μm with a resolution of 512 x 512 pixels and an operating point of 1.5 V were gathered.
Statistical Analysis
Data were saved on EXCEL spreadsheet and statistics were calculated using the Statistical Package for the Social Sciences (SPSS) version 17 (IBM®, Chicago, IL, USA). The Shapiro-Wilk test confirmed the data distribution normality.
The SMH values and the surface microhardness recovery percentage were compared between groups with One-way ANOVA and also the Tukey HSD Post-hoc test. Repeated measures ANOVA and the Sidak Post hoc test were used for within group comparisons. The level of significance was set at 0.05.
Results
Baseline, post lesion and post treatment SMH values and also % SMHR for each group are shown in [Table/Fig-1]. At baseline, SMH values of all samples ranged from 262.40 to 389.30 (mean 330.90±25.06) and no statistically significant differences were seen between groups (p=0.906). Post lesion SMH values ranging from 100.90 to 163.70 (mean 122.88±15.41), were significantly lower than baseline SMH values (p<0.001). However, these values were not significantly different between groups (p=0.209). Post treatment SMH values were significantly different from post-lesion ones (p<0.001) except for control group which was not different statistically (p=0.86). The highest SMH values were observed in NSF group (mean 222.90±28.79) followed by NaF Varnish group (mean 192.48±30.96), n-HAP group (172.65±21.68) and control group (mean 114.11±20.30). Between groups analysis showed statistically significant differences between all groups which is demonstrated in [Table/Fig-2] (p<0.05). However, NaF varnish and n-HAP groups were not different statistically (p=0.165). Recovery of surface microhardness was observed in all interventional groups but % SMHR was negative in control group. Similarly to post treatment SMH values, pairwise comparison of % SMHR revealed significant differences statistically between groups except for NaF varnish and n-HAP groups (p=0.74). The result for % SMHR pairwise comparisonis illustrated in [Table/Fig-2].
Surface microhardness values (mean±SD) and percentage of surface hardness recovery (%SMHR) of groups at baseline, post lesion and post treatment.
| Groups | Baseline | Post lesion | Post treatment | % SMHR |
|---|
| NaF varnish | 327.50±29.34a, A * | 120.81±18.10a, C † | 192.48±30.96b, B | +34.94b |
| n-HAP serum | 333.41±18.29a, A | 126.03±12.71a, C | 172.65±21.68b, B | +22.17b |
| Nano silver fluoride | 329.58±27.36a, A | 116.98±11.74a, C | 222.90±28.79a, B | +50.70a |
| Control | 333.09±26.03a, A | 127.69±17.21a, B | 114.11±20.30c, B | -7.21c |
Different lowercase letters in each column presents statistically significant differences (One-way ANOVA and Tukey post-hoc test p<0.05).
† Different uppercase letters in each row demonstrates statistically significant differences (one sample repeated measure ANOVA and Sidak post-hoc test p<0.05).
Comparison between group of post-treatment surface microhardness values and %SMHR (p<0.05).
| Groups | NaFvarnish | n-HAPserum | Nano SilverFluoride(NSF) | Control |
|---|
| NaF varnish | - | NS | p<0.05 | p<0.001 |
| n-HAP serum | NS | - | p<0.001 | p<0.001 |
| Nano Silver Fluoride (NSF) | p<0.05 | p<0.001 | - | p<0.001 |
| Control | p<0.001 | p<0.001 | p<0.001 | - |
NS presents non-significant difference between groups.
(Post-Hoc Tukey test p<0.05).
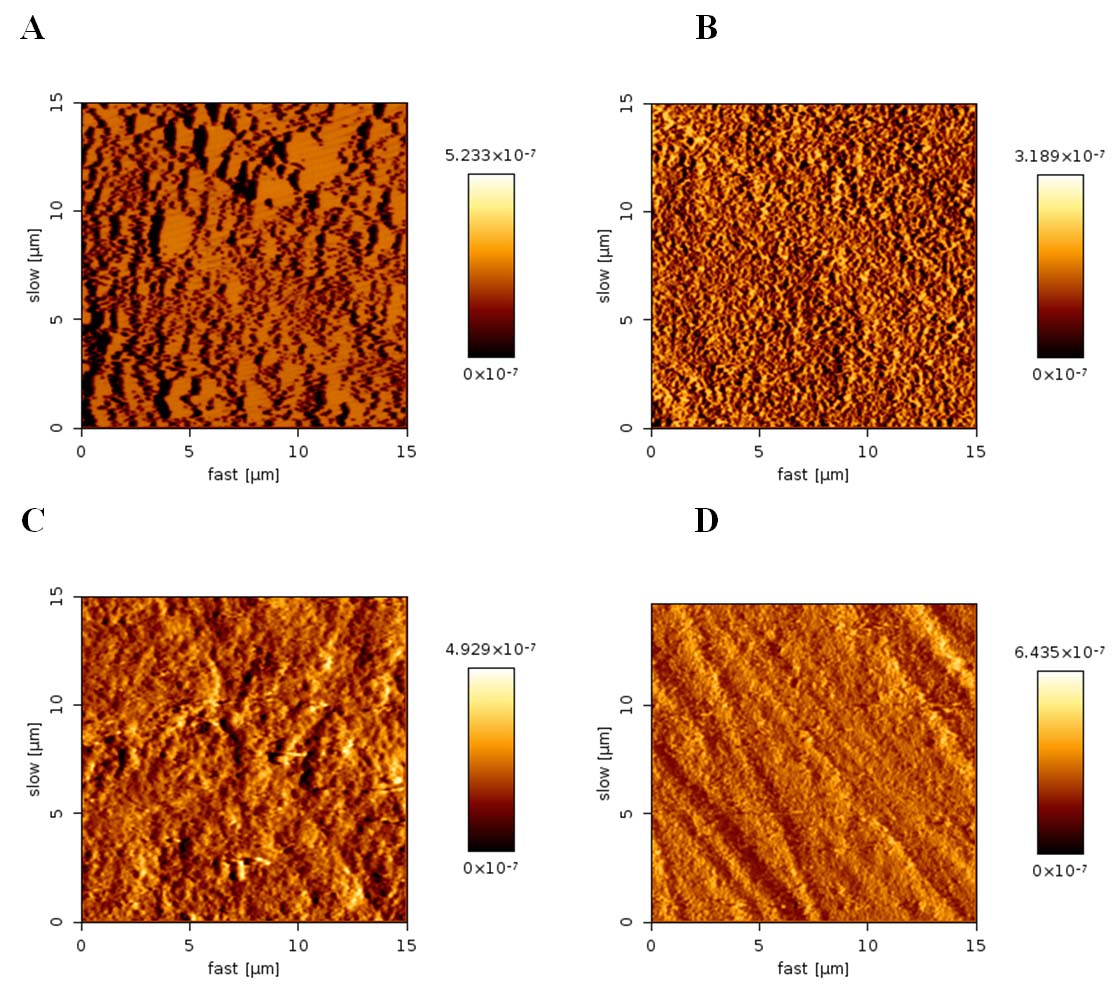
[Table/Fig-3] illustrates diverse AFM images obtained from each group’s enamel surface after remineralization. The NaF varnish group showed development of globular arrangements which were not precisely uniform [Table/Fig-3a]. As it is shown in [Table/Fig-3b], in n-HAP group homogenous acicular crystals were formed. In NSF group, the protective layer consisted of different sized and multi shaped crystal formation [Table/Fig-3c]. No protective layer was observed in the control group [Table/Fig-3d].
AFM images of enamel surfaces after each groups’ remineralization: (A) initial enamel remineralized by NaF varnish, (B) initial enamel remineralized by n-HAP group, (C) initial enamel remineralized by NSF group, (D) No treatment (control group).
Discussion
In this in vitro study, different remineralizing agents were compared to evaluate their effect on demineralized enamel. The null hypothesis which was mentioned earlier was not supported. The results demonstrated that NSF had the highest SMH values. NaF varnish and n-HAP serum also increased the enamel hardness but they were not significantly different.
In the current study, like several previous studies, NaF varnish and n-HAP were effective for enamel remineralization but their comparative efficacy was controversial in reports [6,25,26]. De Carvalho FG et al., compared fluoride varnish and n-HAP paste and similar to our results, both could protect against enamel demineralization. However, in their study n-HAP was more effective than fluoride varnish [26]. This dissimilarity in their findings could be explained by the difference in the n-HAP pastes used in each study. The n-HAP paste applied in the de Carvalho FG et al., study, contained 9000 ppm fluoride while the n-HAP serum in our study did not have any fluoride. This might be the reason to the increased remineralization capability of n-HAP in de Carvalho study.
We applied the n-HAP serum once daily for 10 days. Similar to our results, another study recommended 10% n-HAP to be used daily since its efficacy was similar to 1000 ppm fluoride [27].
In another study conducted by Comar LP et al., fluoride paste (0.2% NaF) was reported to be a better agent in decreasing enamel demineralization in comparison to n-HAP pastes and n-HAP plus fluoride. This result might be due to the different study design. They focused on comparing the prevention of demineralization of the experimental pastes, not the remineralization of the pre formed enamel caries lesion [25].
Tschoppe P et al., reported that n-HAP in the form of toothpaste was more effective than amine fluoride ones in remineralizing enamel and dentin [6]. We used n-HAP (PrevDent International BV, Netherlands) in the form of serum which contained aqua, xylitol, nano-hydroxyapatite, potassium chloride, menthapiperita oil, linalool, and limonene. However, the n-HAP pastes used in Tschoppe P et al., study were in form of toothpaste with different formulations containing n-HAP with and without zinc carbonate. This difference in formulation might describe the different results. Considering fluorosis, the most important fluoride disadvantage in children, n-HAP could be recommended as an alternative. Also, Tschoppe P et al., recommended n-HAP as a substitute to fluoride which is consistent with our results [6].
In the current study, we evaluated the remineralization effectiveness of NSF as a solution. The results showed the highest SMH values and %SMHR. Zhi QH et al., demonstrated that both silver and fluoride ions were responsible for enamel remineralization. As a matter of fact silver ions could infiltrate into carious lesions and precipitate there. This could result in enamel hardening [33].
In the present study, the enamel surfaces were evaluated by AFM and diverse images were obtained as a result of different remineralization mechanisms. All treatment groups formed protective layers which are in agreement with our SMH values results. The granular structure of NaF varnish group was because of CaF2 formation as another research had described [10]. The n-HAP group showed uniform acicular n-HAP crystals which covered the enamel pores. Its organized microstructure seemed like a woven unit protecting the enamel from demineralization attack. The protective layer caused by NSF was heterogeneous and different to the former groups. The diversity in crystals’ sizes and shapes could be explained by their different nano ionic origins. No definite protective layer was observed in the control group except for minor crystals caused by the remineralization solution. The polishing procedures could be the cause to scrape like lines.
Based on the reviewed literatures, this is the first time to evaluate remineralization potential of NaF varnish, n-HAP serum and NSF on enamel carious lesions in primary anterior teeth. As mentioned in dos Santos study NSF is cost-benefit, easy to apply and it could be applied once a year [2]. Therefore, it has the potential to replace NaF varnish as an in-office fluoride therapy substance.
Limitation
In order to better mimic to clinical conditions, we used pH-cycling model. However, in vitro study might not reproduce the results of an in-vivo one. Besides, the effect of oral factors such as, saliva and dental plaque on the enamel remineralization could not be considered. The antimicrobial capacity of silver products was well known but was not the aim of this study. It is also valuable to evaluate the safety of NSF before its clinical application. Therefore, further studies are recommended to evaluate the oral factors, the anti microbial potential and safety in clinical settings.
Conclusion
Based on the results of this in vitro study, NaF varnish and n-HAP serum were similar in remineralizing initial caries. NSF could have the greatest remineralization capability in comparison to other materials evaluated. Therefore, n-HAP serum and NSF can be introduced as substitutes for NaF varnish.
Different lowercase letters in each column presents statistically significant differences (One-way ANOVA and Tukey post-hoc test p<0.05).
† Different uppercase letters in each row demonstrates statistically significant differences (one sample repeated measure ANOVA and Sidak post-hoc test p<0.05).
NS presents non-significant difference between groups.
(Post-Hoc Tukey test p<0.05).