The CS of heart is the largest vein draining the blood from heart. By definition, the CS is the blood conduit in the continuation of the Great Cardiac Vein (GCV) situated between the valve of GCV of Vieussens or the point of entrance of the Oblique Vein of Left Atrium (OVLA) and the Thebesian Valve (ThbV) of the ostium of the CS [1]. It is about 2 cm-3 cm in length, situated in the posterior part of the coronary sulcus of the heart, usually covered by muscular fibers from the Left Atrium (LA) [2]. It continues to run on the posteroinferior region of Atrioventricular groove (AV groove) until it ends at its orifice located between the Inferior Vena Cava (IVC) and the tricuspid valve on the inferior region of the atrial septum. The CS and its tributaries return blood to the Right Atrium (RA) from the whole heart including its septa except the anterior region of the Right Ventricle (RV) and small, variable parts of both atria and Left Ventricle (LV) [3].
The beginning of the CS is marked by either an outer constriction, the opening of the OVLA or by the Vieussens valve internally. The ThbV marks the CS ostium. The terminal part of the CS lies in the upper part of the floor of the inferior pyramidal interatrial fossa, which is an adipose filled region at the crux of the heart containing the U-turn part of the right coronary artery, known as the “U-turn of James” and its atrioventricular branch. The CS is usually cylindrical in shape with the widest diameter towards its opening into the RA. Sometimes it shows marked narrowing at the site of Vieussens valve, giving a funnel shape appearance. The GCV and CS are commonly embedded in the subepicardial fat of the inferior AV groove. The CS as it passes to the right deviates slightly superiorly to become more related to the back of the LA before it courses inferiorly to open into the RA [4].
The CS has become a clinically important structure through its role in providing access for different cardiac procedures. Historically, cardiac vascular studies have focused mainly on the circulation of coronary arteries. The coronary venous system has become important in many electrophysiological procedures, viz. biventricular pacing, arrhythmia ablation and for deployment of an array of cardiac devices [5,6]. The advent of advanced invasive and interventional cardiac treatment and management tools for common disorders like heart failure has made understanding of CS anatomy necessary [7-9]. There is also an increased interest in the CS as an access point for interventionists for ablation procedures as an arrhythmia source and for mapping [10].
Currently one of the most common uses of the cardiac venous system is the placement of left sided pacing leads in cardiac resynchronization therapy. In this, the leads are permanently placed within the cardiac venous system to minimize delayed conduction in the LV. The anatomy of these veins provides a unique access to myocardium of LV [3].
Detailed anatomical knowledge of CS is clinically relevant, but not much data is available on its morphology and morphometry especially in the North Indians. Thus, the present study was conducted to observe various morphological and morphometric parameters of CS in adult human wet specimens of heart.
Materials and Methods
The current cross-sectional observational study comprised of 50 adult human apparently normal formalin fixed cadaveric hearts belonging to either sex (age group 20-80 years). The study was carried out from October 2015 to October 2016 in the Department of Anatomy, Government. Medical College, Amritsar, India. Hearts with congenital anomalies, serious pathology and post surgery evidence were excluded from the study. Dissection was performed with extraordinary care to protect the delicate intrinsic structures of the CS. High resolution pictures were taken with a digital camera (20.1 mega pixel). CS was identified in its normal location, posterior AV groove after removing the fat. It was cleaned along its whole length. Following observations were made and measurements were taken:
1. Presence/Absence: It was noted whether the CS was present or absent;
2. Location: Its location was identified and any deviation from its normal location i.e., left posterior atrioventricular sulcus was noted;
3. Shape: Shape of the CS was determined whether it was tubular, conical, funnel or any other;
4. Length: To measure its External Length (EL) a thread was kept along the length of CS. It was marked at the entry of oblique vein into CS and at the termination of CS into RA. Then it was spread on a flat surface and by using digital vernier caliper, the distance between the two points displayed on vernier caliper was noted and recorded as its EL1 (AB in [Table/Fig-1]). If OVLA was absent, then the marking was made at the outer constriction distal to the valve of Vieussens (EL 2). And if both the oblique vein and valve of Vieussens were not traceable, then the point of union of GCV and Left Marginal Vein (LMV) was used as the landmark (EL 3) (AB in [Table/Fig-2]). The CS wall was sectioned along its long axis to expose its lumen. To measure its Internal Length (IL) a thread was kept along its whole length and marked at the valve of Vieussens and the CS ostium. The distance between these two points was recorded as its IL1 (AB in [Table/Fig-3]). In those specimens where valve of Vieussens was absent the distance between the opening of LMV in the CS and its ostium in RA was measured as its Internal Length (IL 2).
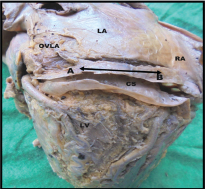
Showing external length of coronary sinus.
AB- external length (EL 1), CS- Coronary sinus, OVLA- oblique vein of left atrium, LA- Left atrium, RA- Right atrium, LV- Left ventricle
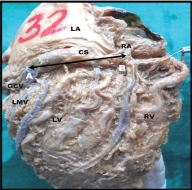
Showing external length of coronary sinus.
AB- external length (EL 3), CS- Coronary sinus, LA- Left atrium, RA- Right atrium, LV- Left ventricle, RV- Right ventricle
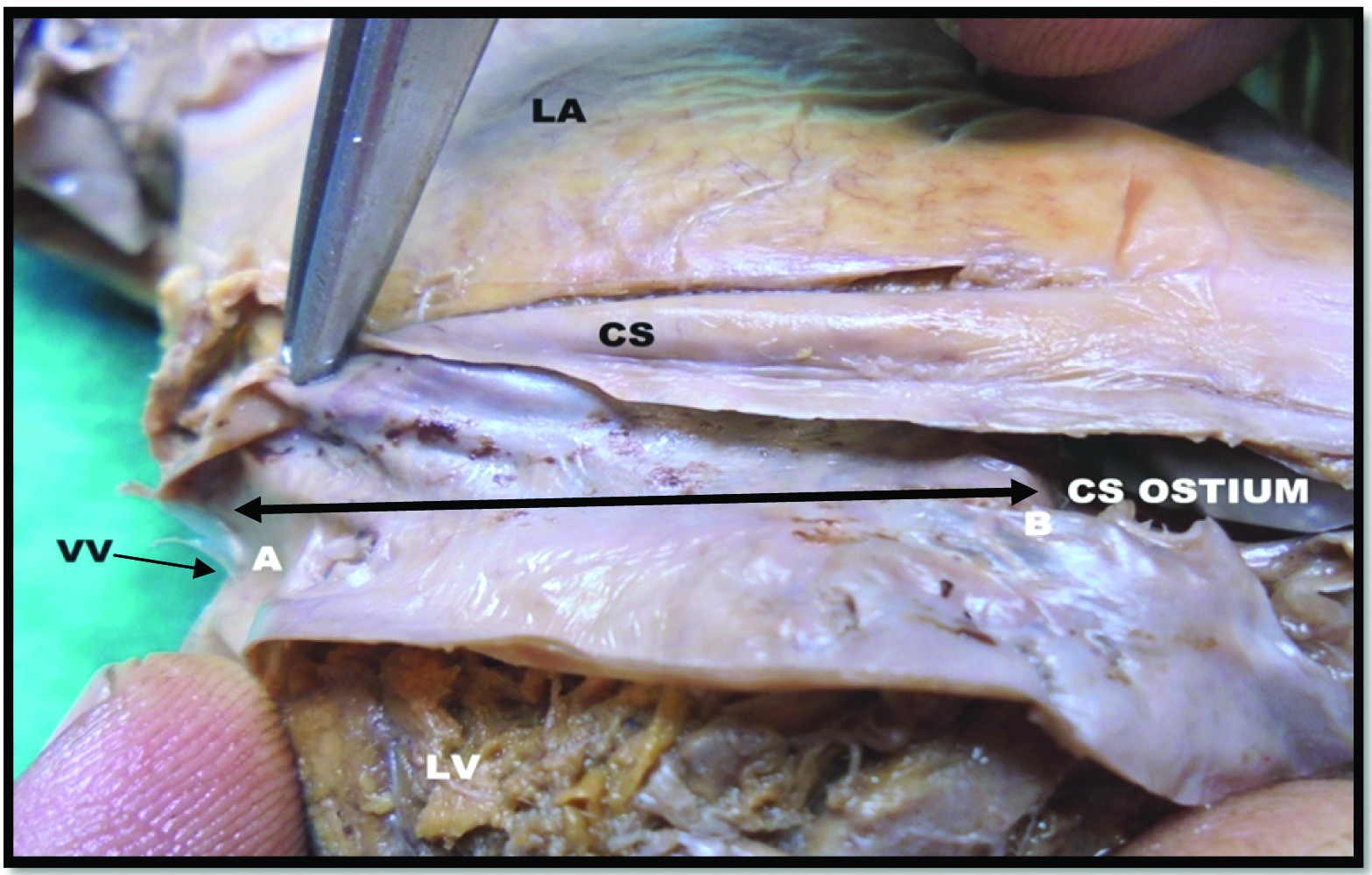
Showing internal length (IL) of coronary sinus.
(AB-internal length, CS-coronary sinus, VV-valve of Vieussens, LA-left atrium and LV-left ventricle).
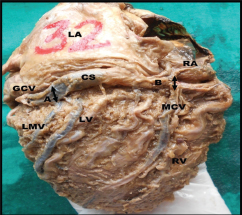
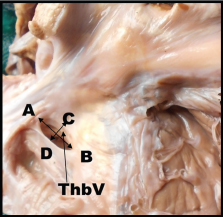
5. Width: The width of CS was measured with the help of a divider and vernier caliper at three levels; at the beginning (A in [Table/Fig-4]) at the point of entry of MCV (B in [Table/Fig-4]) and at its termination in RA. Maximum and minimum width of CS at its termination was noted separately. Maximum width measured parallel to the free edge of valve, excluding the valve (AB in [Table/Fig-5]) and minimum width was measured from the free edge of valve to the opposite margin of CS at its termination (CD in [Table/Fig-5]).
Showing width of Coronary Sinus (CS) at its beginning (A) and at the entry of middle cardiac vein (MCV); (B) GCV- great cardiac vein, LMV-left marginal vein, LA-left atrium and LV-left ventricle, RA-right atrium, RV-right ventricle
Showing width of coronary sinus at its termination. AB-maximum width, CD-minimum width, ThbV-Thebesian valve.
6. Muscles of left atrium covering the coronary sinus: The walls of CS were carefully examined to note whether it was covered by the muscles of left atrium or not. If found to be covered with muscles then it was noted that how much length of CS was covered whether one-third, one-third to two-third or more than two-third.
Statistical Analysis
Descriptive analysis (based on One-way ANOVA test and Pearson correlation test) was done to calculate range, mean and SD by using SPSS software version 17.0.
Results
CS was present in all the 50 hearts studied in present study. A constant location of CS in the left part of posterior AV sulcus or coronary sulcus was observed in 49 hearts (98%). However, in one case it was located 6.8 mm above the posterior AV groove [Table/Fig-6].
Mainly two shapes of CS were observed in present study viz., tubular shape in 41 hearts (82%) where there was gradual tapering off of the distal segment [Table/Fig-7] and funnel shape in 9 hearts (18%) where sudden tapering off of the distal segment [Table/Fig-8] was seen.
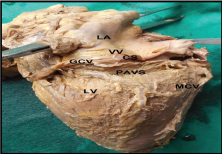
Showing location of coronary sinus (CS) above the posterior atrioventricular sulcus (PAVS). (GCV- Great cardiac vein, LA- left atrium, LV- left ventricle, VV- valve of Vieussens, MCV- middle cardiac vein).
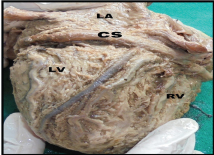
Showing tubular/cylindrical shaped Coronary Sinus (CS).
LA- left atrium, LV- left ventricle, RV- right ventricle.
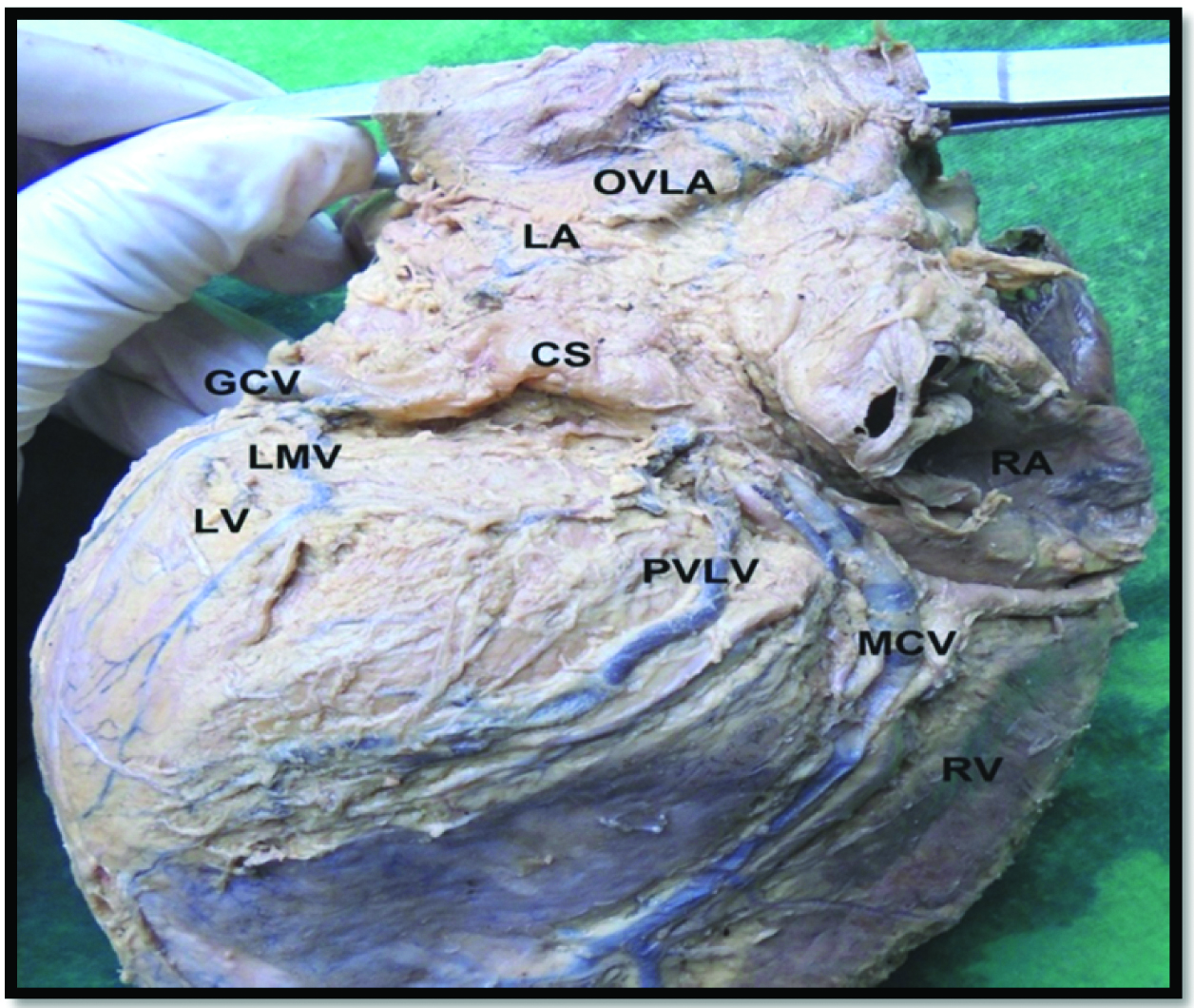
Showing funnel shaped Coronary Sinus (CS).
(GCV- great cardiac vein, LA- left atrium, LV- left ventricle, OVLA – Oblique vein of left atrium, LMV- Left marginal vein, MCV- middle cardiac vein, PVLV- Posterior vein of left ventricle).
The total external length of CS (EL) irrespective of the landmark used, ranged from 20.5 mm to 58.78 mm, mean being 38.22±8.6 mm. The total IL irrespective of landmark used ranged from 16.28 mm to 49.6 mm; mean being 34.48±8.9 mm. Mean and SD of external and internal length were calculated separately for different landmarks used, as shown in [Table/Fig-9]. EL was found to be more than IL. It was maximum when measured from the outer constriction distal to valve of Vieussens to termination of CS (38.7±9.01). The difference between EL and IL was found to be significant statistically (Pearson correlation; r=0.803, p<0.001).
Length of the coronary sinus.
| S. No | Length | No. of specimens | Mean±SD (mm) | Range(mm) |
|---|
| 1 | EL 1 (between OVLA and termination of CS) | 20 | 38.45±8.32 | 20.50 - 57.12 |
| 2 | EL 2 (between outer constriction distal to valve of Vieussens and termination of CS) | 26 | 38.70±9.01 | 20.58 - 58.78 |
| 3 | EL 3 (from the point of union of GCV and LMV to termination of CS) | 4 | 33.81±8.7 | 21.35 - 40.72 |
| 4 | EL (total) | 50 | 38.22±8.6 | 20.50 - 58.78 |
| 5 | IL 1 (between valve of Vieussens and CS ostium) | 44 | 35.72±8.6 | 18.76 - 49.6 |
| 6 | IL 2 (between LMV ostium and CS ostium) | 6 | 25.45±6.9 | 16.28 - 33.92 |
| 7 | IL (total) | 50 | 34.48±8.9 | 16.28 - 49.6 |
The width of CS was measured at three different levels; at its beginning, at the entry of MCV into CS and at its termination in RA. Values obtained are shown in [Table/Fig-10]. Width of CS increased towards its termination, minimum (4.25±1.4) was at its beginning and maximum (9.61±2.6) at its termination in RA. There was a statistical significance between the width at beginning and at the entry of MCV (r=0.543; p<0.001) and also between maximum and minimum width, at its termination in RA (r=0.430; p=0.002).
Width of the coronary sinus at different level.
| S. No. | Level | Mean±SD (mm) | Range (mm) |
|---|
| 1 | At beginning | 4.25±1.4 | 2.25-8.32 |
| 2 | At entry of MCV | 6.98±2.7 | 2.4-13.9 |
| At termination in RA |
| Maximum | 9.61±2.6 | 4.02-16.40 |
| Minimum | 5.79±2.1 | 2.30-10.00 |
In 96% of hearts CS was covered by muscles of LA [Table/Fig-11]. Values are given in [Table/Fig-12].
Showing muscle of left atrium (LA) covering coronary sinus (CS).
LV- left ventricle, RV- right ventricle, MCV- middle cardiac vein.
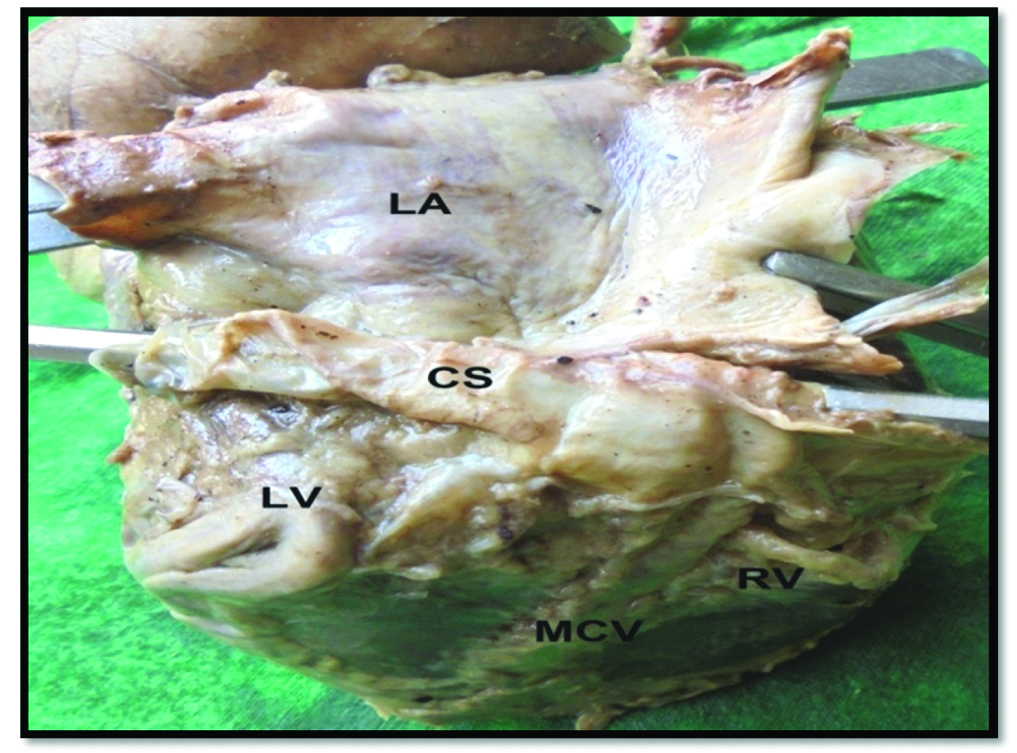
Muscles of left atrium covering coronary sinus.
| S. No | Muscles of LA | No of specimens (%) |
|---|
| 1 | Covering >2/3rd of CS | 46 (92) |
| 2 | Covering1/3rd to 2/3rd of CS | 2(4) |
| 3 | Covering <1/3rd of CS | 0 (0) |
| 4 | No muscle cover for CS | 2 (4) |
| Total | 50 (100) |
Discussion
A complete CS was present in all cases, consistent with literature reports that absence or the unroofed variants are rare [11-14]. CS was located in the left part of posterior AV groove or coronary groove. It was comparable to Ominde BS et al., [15]. But in one heart of present study, it was located on the posteroinferior part of the LA 6.8 mm above the posterior AV groove as seen in [Table/Fig-6]. It was a rare finding which could not be traced in the accessible literature. This abnormal position of CS on the posterior wall of LA may lead to compression of CS in case of left atrial enlargement.
The incidence of the shapes of CS is compared in [Table/Fig-13] [4,15-17]. The shape of CS in the present study was comparable to previous studies [4,16] except Ominde BS et al., and Karagoz A et al., they found wind-sock shape (similar to funnel shape) more frequently [15,17]. This shows that shape of CS may vary in different population. It is also found that a cylindrical or tubular intracoronary device is more effective than a funnel device [4,16].
Showing comparison of shape of the coronary sinus.
| S. No | Authors | Population (Year) | Shape | % | No. of Cases |
|---|
| 1 | El-Massarany S et al., [4] | British (2005) | Cylindrical | 80 | 32/40 |
| Funnel | 20 | 8/40 |
| 2 | Bellasteros LE et al., [16] | Colombian (2010) | Cylindrical | 67.6 | * |
| Funnel | 23.5 | * |
| Flattened | 8.9 | * |
| 3 | Karagoz A et al., [17] | Turkish (2013) | Wind-sock | 56.2 | 9/16 |
| Tubular | 43.8 | 7/16 |
| 4 | Ominde BS et al., [15] | Kenyan (2015) | Wind-sock | 100 | 74/74 |
| 5 | Present study | Indian (2016) | Tubular/ cylindrical | 82 | 41/50 |
| Funnel | 18 | 9/50 |
* Not available.
[Table/Fig-14] shows a comparison of length of CS as observed by different workers in different population and by different methods [4,11,12,15,16,18-27]. It is seen that the mean length of CS varies between 25.96 mm and 109 mm in different studies.
Showing the comparison of length of the coronary sinus [4,11,12,15,16,18-27].
| S. No | Authors | Year | Population (no of cases) | Length | Method |
|---|
| Mean(mm) | Range(mm) | |
|---|
| 1 | El-Massarany S et al., [4] | 2005 | British (32) | 48.4±5.2 | * | Cadaveric |
| 2 | Lee MS et al., [18] | 2006 | American (*) | 86.5 | * | Venography |
| 3 | Plass A et al., [19] | 2008 | Swiss (50) | 108.9±18 | * | CT |
| 4 | Habib A et al., [20] | 2009 | American (*) | * | 30-55 | Cadaveric |
| 5 | Ballesteros LE et al., [16] | 2010 | Columbian (68) | 25.96±6.34 | * | Autopsy |
| 6 | Sun JP et al., [21] | 2012 | American (*) | 109 | * | MSCT |
| 7 | Ankolekar VH et al., [12] | 2013 | S/Indian (55) | 28 | 20-38 | Cadaveric |
| 8 | Zhaoming Z [22] | 2013 | Chinese (*) | 38.7 | * | Cadaveric |
| 9 | Suma HY et al., [11] | 2013 | S/Indian (25) | * | 20-55 | Cadaveric |
| 10 | Noheria A et al., [23] | 2013 | *(620) | 40 | * | Autopsy |
| 11 | Kavimani and Jebkani CF [24] | 2014 | S/Indian (40) | 28 | 20-38 | Cadaveric |
| 12 | Manoranjitham R et al., [25] | 2015 | S/Indian (30) | 54.98±12.2 | * | Cadaveric |
| 13 | Loch WB et al., [26] | 2015 | Polish (80) | * | 15-50 | Cadaveric |
| 14 | Ominde BS et al., [15] | 2015 | Kenyan (74) | 39.55±5.32 | 20-53 | Cadaveric |
| 15 | Mehra L et al., [27] | 2016 | N/Indian (40) | 35.35±4.43 | 20.89-39.62 | Cadaveric |
| 16 | Present study | 2016 | N/Indian (50) | | | Cadaveric |
| EL | | | EL 38.22±8.6 | 20.5-58.78 |
| | EL 138.45±8.3 | 20.50-57.12 |
| | EL 238.7±9.01 | 20.58-58.78 |
| | EL 333.81±8.7 | 21.35-40.72 |
| IL | | | IL34.48±8.9 | 16.28-49.6 |
| | IL 135.72±8.6 | 18.76-49.6 |
| | IL 225.45±6.9 | 16.28-33.92 |
* Not available, S/Indian- South Indian, N/Indian-North Indian
Such a wide range of length of CS as seen in above mentioned studies [Table/Fig-14] may be attributed to differences in methodology, population and stature which all may affect the size of heart and its vessels. Sun JP et al., who studied CS in American population by Multi Slice Computed Tomography (MSCT) method revealed a higher value [21] while Lee MS et al., in the same population using venography obtained a different result [18]. This shows that the length of CS varies with methodology even in the same population. It is evident that despite the above mentioned factors which could affect the length of CS, it also varies due to the different landmarks used; from Vieussens valve to CS ostium [4], from the point of entry of OVLA to CS ostium, from the union of GCV and LMV up to the CS ostium [25], from the union of GCV and OVLA to CS ostium [15] etc.
In the present study EL 1 was measured from the point of entry of OVLA into CS up to the junction where CS pierces the wall of RA in 20 hearts (40%). Same landmark was used by Suma HY et al., and Ominde BS et al., and our values are in line with them [11,15]. In 26 cases (52%) EL 2 was measured from the outer constriction just distal to the valve of Vieussens to the CS ostium. There is no data available for comparison. In remaining four cases (8%) external length was measured from the union of GCV and LMV to the CS ostium (EL 3) and our values were found to be low as compared to that of Manoranjitham R et al., who also used the same landmark. IL1 of CS [25], measured from the valve of Vieussens to the CS ostium in 44 hearts (88%) of the present study was lower than the values of El-Maasarany S et al., who also used same land mark for measuring the length of CS [4]. In remaining six hearts (12%) internal length was measured from the LMV ostium in CS to the CS ostium. There was no data available for comparison. Comparatively lower values were found in autopsy as well as cadaveric studies. Hence, the present study is comparable to Ominde BS et al., Zhaoming Z and Mehra L et al., as seen in [Table/Fig-14] [22,15,27].
Since, the length of CS is important in designing of cannulation devices used in cardiac resynchronization therapy and percutaneous mitral valve annuloplasty, length of CS is obtained both externally and internally. These two measurements matter when a cardiac surgeon has to choose a vein for catheterization or mapping. If OVLA is chosen then EL has to be considered or if it is GCV then IL is considered. However, it displays gender and population variations that may account for the failure rate of these procedures [15]. It was observed that width of CS increased towards its termination. In case of CS hypoplasia the device inserted into the CS might obstruct its lumen; especially its narrow distal part.
Chauvin M et al., found that there were connections of striated myocardium of LA of varying number and morphology with coronary muscle cuff; they ranged from 1 to 2 fascicles to a widely intermingled continuum [28]. In 96% of hearts of present study CS was covered by left atrial musculature and in 92% cases, the coverage has extended more than its 2/3rd portion. It may increase the risk of arrhythmia because Katritis DG et al., believes that atrial myocardial extensions into cardiac venous structures provide the substrate for potential arrhythmogenecity [29].
Although, CS is a constant structure in cardiac venous anatomy, its location is not always the same. A rare location of CS on the posteroinferior wall of LA observed in this study can lead to CS compression in left atrial enlargement. Shape and width of CS are important parameters to look for before designing an intracoronary sinus device. The range of length in present study exceeded the range of 2 cm-3 cm documented in the standard anatomy text book. So, it may be possible that during the cannulation of CS with a standard device the whole length of CS could not be visualized and this could be a reason for the failure of implantation of pacing leads.
Limitation
This study does not provide the knowledge about gender and age differences in the anatomy of CS, so gender preponderance of CS cannot be defined. Further more accurate anatomical studies can be done using advanced radiological techniques such as MSCT in order to achieve optimal results in the future. However, more about the morphology of CS needs to be explored as it is a potential site for advanced clinical and translational research.
Conclusion
Estimation of length of CS is helpful to design or select devices of appropriate length in order to make cannulation easy and less time consuming. The left atrial muscle coverage of CS may be associated with increased chance of developing arrhythmogenecity. This study may provide a baseline data which can be of help to conventional and interventional cardiologists.
* Not available.
* Not available, S/Indian- South Indian, N/Indian-North Indian