Non-Catheter Related Perioperative Superior Vena Cava Syndrome Following Surgical VSD Closure-Role of Transesophageal Echocardiography
Tanveer Singh Kundra1, Divya Gopal2, Manasa Dhananjaya3
1 DM Resident, Department of Cardiac Anaesthesia, Sri Jayadeva Institute of Cardiovascular Sciences and Research, Bengaluru, Karnataka, India.
2 Assistant Professor, Department of Cardiac Anaesthesia, Sri Jayadeva Institute of Cardiovascular Sciences and Research, Bengaluru, Karnataka, India.
3 Assistant Professor, Department of Cardiac Anaesthesia, Sri Jayadeva Institute of Cardiovascular Sciences and Research, Bengaluru, Karnataka, India.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Dr. Tanveer Singh Kundra, Kothi No.184, Phase 4, Mohali, Punjab-160055, India.
E-mail: tvskundra@yahoo.co.in
Unintentional iatrogenic surgical complications can complicate the operative and postoperative courses of paediatric cardiac surgery patients. Unless recognized and treated early, it’s possible, these complications may lead to a prolonged hospital stay, increased treatment cost, morbidity, and even death. Ventricular Septal Defect (VSD) is the most common congenital heart defect in children, occurring in 50% of all children with congenital heart disease and in 20% as an isolated lesion. Herein, we discuss the development of Superior Vena Cava (SVC) syndrome following surgical repair of VSD in a nine-month-old child. Early clinical diagnosis, immediate confirmation with Transthoracic Echocardiography (TTE) and surgical re-exploration led to a good outcome in this patient. If, however, we could have performed an intraoperative Trans Esophageal Echocardiography (TEE), this complication could have been recognized, even before shifting from the operating room.
Iatrogenic, Surgical complication, Upper torso discoloration
Case Report
A nine-month-old child weighing 5.5 kg was admitted for surgical VSD repair. Surgery went off as scheduled. While trying to cannulate the left femoral artery, the left femoral vein was punctured and we decided to retain the left femoral venous access for intravenous fluids and medications, instead of cannulating the right Internal Jugular Vein (IJV). Right femoral artery was cannulated for haemodynamic monitoring. Haemodynamics of the patient were monitored in the form of heart rate and invasive blood pressure, which were stable throughout the procedure. Central Venous Pressure (CVP) was also monitored, but since femoral vein was cannulated, the trend was more important than the absolute value, which remained normal throughout the procedure. VSD patch closure was done. Post-procedure step-up (i.e., difference between right atrial saturation and pulmonary arterial saturation) was 9% and pulmonary artery pressures were <1/3 systemic pressures.
While removing the SVC cannula in the patient, there was bleeding from this site. Suturing of the bleeding site was done. Haemodynamics were maintained with fluids. Haemostasis was achieved and after closing the chest, the patient was shifted to the postoperative recovery room.
Immediately on shifting to the recovery room, it was observed that the child had progressively increasing congestion and discoloration of the upper body. First, it involved the face, that further spread to the upper torso [Table/Fig-1]. There was swelling of the face and particularly eyelids. However, haemodynamics were stable at this point of time. CVP through the femoral line was also normal. However, the Arterial Blood Gas (ABG) test showed an increased lactate level of 5.7 mmol/l.
Congestion and discoloration involving the face and the upper torso, giving rise to clinical suspicion of SVC Syndrome;
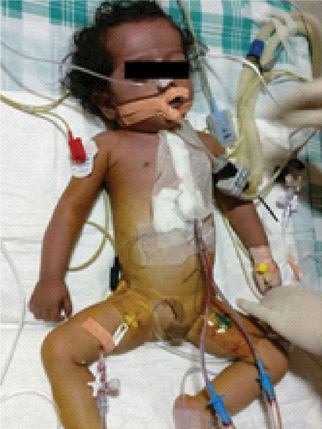
A clinical suspicion of SVC syndrome was made and immediately a TTE was done. TTE showed very sluggish flow in the SVC.
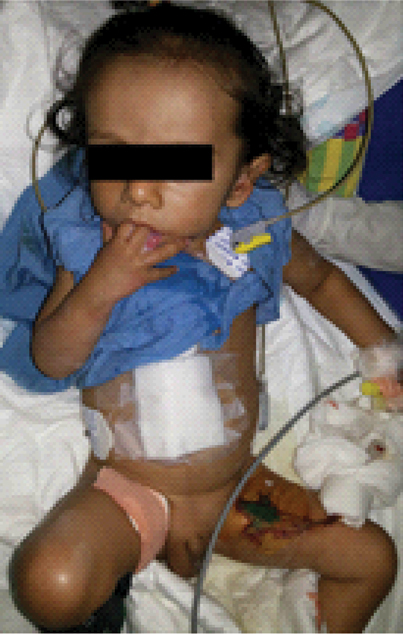
An immediate decision was made to re-explore the child. On opening the chest, there was a blood clot sitting on the SVC. Also, it was decided to repair the SVC as the diameter seemed smaller at the site of suturing. Hence, a shunt was made between the innominate vein and the Right Atrium (RA). SVC was clamped and the sutures were opened. Pericardial patch augmentation of SVC was done. Immediately post-procedure, the discoloration and oedema of the face disappeared. Eyelids seemed normal and the lactates reduced to 2.5 mmol/L in the ABG test. The arterial lactates further decreased to 1.3 mmol/L in the postoperative ABG test in the recovery. The baby was subsequently extubated uneventfully and had a good outcome despite the surgical complication [Table/Fig-2].
Child was extubated next day after surgery and had a good outcome.
Discussion
VSD is the most common congenital heart defect in children with an incidence range of 1.56-53.2/1,000 live births [1].
Acute intraoperative SVC syndrome is a very rare complication in patients undergoing cardiac surgery. Although, catheter related SVC syndrome has been previously reported, but to our knowledge, this is one of the very few cases reported of non-catheter related acute intraoperative SVC syndrome in the cardiac surgical patient population [2]. Since, acute intraoperative SVC syndrome is rarely seen, its management has not been well-documented.
The management of acute SVC syndrome involves finding and relieving the cause of SVC obstruction. The most common causes of acute iatrogenic SVC syndrome are thrombus formation and SVC stenosis and in any such patient these causes must be excluded [3].
Secondly, echocardiography can play an invaluable role in diagnosing SVC obstruction as well as identifying the site of obstruction. If the point of obstruction cannot be relieved fast, haemodynamic instability may occur as the result of a marked decrease in preload.
Thirdly, intravenous access should then be changed to the lower extremity veins (i.e., femoral veins), and all fluids and medications should be given through these lines to allow them to reach the systemic circulation. In our patient, venous access was already in the left femoral vein.
Lastly, SVC obstruction and the resultant rise in CVP and intracranial pressure can also cause a decrease in cerebral perfusion pressure and can lead to cerebral oedema. Hence, the obstruction needs to be relieved as early as possible [3].
Reverse trendelenberg position aids in venous drainage. Hence, in a patient of suspected SVC syndrome, patient positioning may also be helpful. Diuretics are not very helpful in acute SVC syndrome because their effects are delayed [4].
Collateral venous drainage takes several weeks to make a significant impact. Hence, collaterals do not play a role in a case of acute SVC obstruction, like in the present case [4-6].
The incidence of non-carcinogenic causes of SVC Syndrome is on the rise [7]. However, there have only been a few case reports of acute SVC syndrome in the cardiac surgical patient population [8,9]; even fewer reports are there for non-catheter related acute SVC syndrome [3].
The above case report highlights the importance of anticipating surgical complications, their early recognition by a good clinical examination, the importance of perioperative echocardiography, especially intraoperative TEE, in such patients and the willingness on the part of both surgical as well as an anaesthetic teams to immediately re-explore the patient as soon as the diagnosis is made.
This type of complication would have been easily identified by the use of TEE before leaving the Operating Room (OR). But since a paediatric TEE probe was not available, hence no TEE was performed on this patient after coming off cardiopulmonary bypass. If we perform a routine TEE for all patients before chest closure, such causes of re-exploration could be avoided. The complication could have been life-threatening if it was not recognized in time.
Summarizing, we present an emergency case of SVC Syndrome caused by vary tight suturing of the SVC following SVC cannula removal after Cardio-Pulmonary Bypass (CPB) in VSD closure surgery. In addition, the situation was worsened by a blood clot sitting on the SVC. The condition was recognized early due to clinical suspicion and echocardiography. The complication could have been recognized before leaving the OR, had a TEE been performed on the patient.
[1]. Minette MS, Sahn DJ, Ventricular septal defectsCirculation 2006 114:2190-97. [Google Scholar]
[2]. Kumar R, Iatrogenic superior vena cava syndrome during off pump CABGAnn Cardiac Anaesth 2012 15:320-21. [Google Scholar]
[3]. Amundson AW, Pulido JN, Hayward GL, An unusual cause of intraoperative acute superior vena cava syndromeAnn Card Anaesth 2013 16:133-36. [Google Scholar]
[4]. Wilson LD, Detterbeck FC, Yahalom J, Clinical practice. Superior vena cava syndrome with malignant causesN Engl J Med 2007 356:1862-69. [Google Scholar]
[5]. Kim HJ, Kim HS, Chung SH, CT diagnosis of superior vena cava syndrome: Importance of collateral vesselsAJR Am J Roentgenol 1993 161:539-42. [Google Scholar]
[6]. Trigaux JP, Van Beers B, Thoracic collateral venous channels: Normal and pathologic CT findingsJ Comput Assist Tomogr 1990 14:769-73. [Google Scholar]
[7]. Rice TW, Rodriguez RM, Light RW, The superior vena cava syndrome: clinical characteristics and evolving etiologyMedicine 2006 85:37-42. [Google Scholar]
[8]. Maggiano HJ, Higgins TL, Lobo W, Makos G, Golding LA, Superior vena cava syndrome after open heart surgeryCleve Clin J Med 1992 59:93-95. [Google Scholar]
[9]. Sze DY, Robbins RC, Semba CP, Razavi MK, Dake MD, Superior vena cava syndrome after heart transplantation: percutaneous treatment of a complication of bicaval anastomosesJ Thorac Cardiovasc Surg 1998 116:253-61. [Google Scholar]