CrGN is defined as any glomerular disease characterized by extensive crescents involving more than 50% of the glomeruli with a rapid loss of renal function (at least 50% decline in the glomerular filtration rate {GFR} within 3 months). It includes pauci-immune, immune complex-mediated and anti-glomerular basement membrane disease. It is one of the leading causes for acute or rapidly progressive renal failure. The prognosis is poor with CrGN with an increased risk of end stage renal disease and death despite immunosuppressive therapy. There is regional and temporal variation in aetiology, prevalence and prognosis of diffuse CrGN across the world [1-5]. There is limited data on the aetiology, clinico pathological spectrum and outcomes of CrGN in adult Indian population. This study aims to assess clinicopathological features and outcome of diffuse CrGN in Southern India.
Materials and Methods
In this retrospective study, we reviewed clinical records of all adults (>18 years) admitted to Kasturba medical college, Manipal University, Manipal, India, with a histopathological diagnosis of CrGN over a 5-year period (Jan 2010-Dec 2014). CrGN was defined as presence of proliferation of parietal cells forming two or more cell layers filling the bowman’s space in more than 50% glomeruli in the renal biopsy [4]. Inclusion criteria: The biopsy proven cases of CrGN where >50% glomeruli showed crescents were included.
Exclusion criteria: Patients with focal crescents (<50% of glomeruli) are excluded from the study.
Those cases that have insufficient data are also excluded. The clinical, biochemical, serological, and histopathological data were collected from the case records in detail for all the patients. The clinical details included were age, gender, duration of the disease, hypertension, edema, oliguria, treatment regimens given, requirement of haemodialysis and the follow-up data for six months.
The serum creatinine at admission, maximum creatinine during the illness and on follow-up was collected for all patients. Urine routine and microscopy, urine protein creatinine ratio or 24 hour protein levels were recorded. The serological results for anti-nuclear antibody (ANA), Anti- Neutrophil Cytoplasmic Antibody (ANCA), anti-double stranded DNA (ds DNA), Anti-Glomerular Basement Membrane antibodies (Anti-GBM) and serum complement levels were collected.
The histological evaluation included total number of glomeruli, number (and proportion) of glomeruli with crescents number (and proportion) with cellular/fibrocellular/fibrous crescents, presence of tuft necrosis, presence of rupture of bowman’s capsule, degree of Interstitial Fibrosis and Tubular Atrophy (IFTA), presence of arteriolar fibrinoid necrosis, interstitial infiltrates and acute tubular necrosis. Immunofluorescence pattern, positivity and intensity were recorded.
Outcomes studied were renal recovery/loss and mortality at the end of six month follow up. The complete recovery was defined as normal urinalysis and estimated GFR >90 mL/min/1.73 m2). The partial recovery was defined as estimated GFR >60 mL/min/1.73 m2 with presence of abnormal urinalysis: microscopic hematuria, 1+ proteinuria with or without RBC casts. The risk factors associated with renal loss and mortality were also studied.
Statistical Analysis
The normally distributed data such as age and number (no.) of glomeruli were presented as mean and standard deviation. The skewed data such as serum creatinin, obsolescent glomeruli, cellular crescents and fibrous/fibro cellular were presented as median and Interquartile Range (IR). The categorical variables were presented as frequency with percentage. Chi-square test was used for analysing the data of outcome versus final diagnosis. The univariate analysis was performed to identify the predictors of renal loss and mortality with a p-value of <0.05 was considered as statistically significant. Data was analysed using SPSS version15.
Results
There were twenty nine cases of diffuse CrGN accounting for an incidence of 2.9% among 1016 non-transplant kidney biopsies in five years. The most common cause was pauci-immune crescentic GN (41%) followed by immune complex (28%), Anti-GBM disease (24%) and one had both Anti-GBM and ANCA positivity (3.5%). There was one case of Thrombotic Micro Angiopathy (TMA) with crescents due to malignant hypertension (3.5%) [Table/Fig-1].
Distribution of Diffuse Crescentic Glomerulonephritis as per aaetiology.
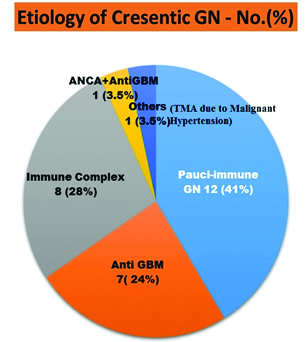
The mean age of the study population was 42.52±17.27 years and 44.8% were females. The relevant demographic, clinical and biochemical features are shown in [Table/Fig-2].
Demographic and clinical characteristics of CrGN.
| Characteristic | All (n=29) (%) |
|---|
| Age (years) ‡ | 42.52 ± 17.27 |
| SCr † at admission * (mg/dl) | 7.2 (IR 3.3 - 10.4) |
| SCr † (maximum) * | 9.0 (IR 5.0 - 11.45) |
| Females† | 13 (44.8) |
| Oliguria† | 9 (31) |
| Edema† | 23(79.3) |
| Hypertension† | 24 (82.8) |
| Nephrotic syndrome† | 4 (13.8) |
| Requirement of Haemodialysis† | 22 (75.9) |
| Microscopic hematuria† | 29 (100) |
| Fever† | 11 (37.9) |
| Rash† | 5 (17.2) |
| Arthralgia† | 5 (17.2) |
*Data represented in Median (I R- Inter-quartile range), †- %- Percentage, ‡ (mean ± SD), SCr-serum creatinine
Four patients presented with nephrotic range proteinuria. All patients had microscopic hematuria and 82.8% of patients had hypertension at presentation. The oliguria was found in 31% of the patients and the average serum creatinine at admission was 7.2 mg/dl (IR 3.3 - 10.4). The dialysis was required in 75% of the patients.
Histopathology features are shown in [Table/Fig-3], 47.5% of the patients had cellular crescents at presentation and 23% had fibrous/fibro cellular crescents. No patients had arteriolar fibrinoid necrosis. Significant interstitial fibrosis and tubular atrophy (>25%) was seen only in 10% of the cases [Table/Fig-3].
Histopathological characteristics.
| Characteristic | All (n =29) (%) |
|---|
| No. of Glomeruli# | 13.24 ± 4.95 |
| Obsolescent Glomeruli %* | 6.66 (IR 0- 15.4) |
| Crescents %* | 77 (IR 60.55 - 90) |
| Cellular crescents %* | 47.05 (IR 24.8- 64.1) |
| Fibrous/Fibro cellular%* | 23.07 (IR 0 - 51.6) |
| Endocapillary-proliferation† | 5 (17.2) |
| Mesangial proliferation† | 11 (37.9) |
| Neutrophil infiltration† | 4 (13.8) |
| Tuft Necrosis† | 8(27.6) |
| Bowman’s capsule rupture† | 10 (34.5) |
| Arteriolar fibrinoid necrosis† | 0 (0) |
| IFTA (>25%)† | 3 (10.34) |
| ATN† | 11 (37.9) |
| Interstitial infiltrates† | 22 (75.9) |
**Data represented in Median (IR- Inter-quartile range), †- %- Percentage, IFTA-Interstitial fibrosis tubular atrophy, ATN-Acute tubular necrosis, SCr-serum creatinine
All the patients received standard protocol based immunosuppression in addition to supportive care. Plasmapheresis was given to subgroup of Anti-GBM disease and ANCA vasculitis as per recommendation. The patients who were declared as End Stage Renal Disease (ESRD) had received immunosuppression for at least one to three month duration.
Among 29 patients, 8 patients died accounting for 27.6% of mortality in our study at the end of six months. Two patients lost to follow-up. At the end of follow-up 34.5% patients had complete/partial recovery and 30% has reached end stage renal disease requiring maintenance haemodialysis [Table/Fig-4].
Outcome at 6 month follow-up.
| Outcomes | All (n =29) (%) |
|---|
| Complete Recovery | 2 (6.9) |
| Partial recovery | 8 (27.6) |
| CKD 5D | 9 (31) |
| Mortality | 8(27.6) |
| Lost for follow-up | 2 (6.9) |
In our study, there were seven Anti-GBM disease patients and all had positive Anti-GBM titers. Two patients had associated pulmonary haemorrhage and rest had renal limited disease. Two patients had partial recovery with a creatinine of 1.8 mg/dl and 2.3 mg/dl at the end of six months. Both patients had a creatinine <5.5 mg/dl and were non-oliguric at presentation. One patient expired and 4 patients reached ESRD. One patient had both Anti-GBM and ANCA positivity and he also succumbed to death due to sepsis.
In immune Complex CrGN subgroup (N=8), the most common was IgA with CrGN (4) followed by PIGN (2), Lupus (1) and MPGN (1) respectively. In this subgroup the prognosis was good. Six patients had complete/partial recovery and one patient reached end stage renal disease and one was lost to follow-up and there was no mortality.
In our study, among pauci-immune CrGN (N=12) patients, ANCA positivity was found in six patients (3 were p-ANCA and three were c-ANCA) and six had ANCA negative status. Irrespective of ANCA status, the prognosis was poor with six patients died and three patients became ESRD and one patient lost to follow-up.
One patient in our study had malignant hypertension with thrombotic microangiopathy and he did not recover and reached ESRD and continued on maintenance haemodialysis.
The univariate analysis was performed to identify the predictors of renal loss and mortality. The significant risk factors associated with renal loss and mortality on univariate analysis were oliguria (p=0.02), requirement of haemodialysis at presentation (p=0.001) and high serum creatinine at admission (>5.5mg/dl) (p=0.003). Histopathological features like presence of fibrous/fibrocellular crescents (p-0.34) or IFTA (p-0.23) did not influence the outcome in our study.
Discussion
The incidence of CrGN varies with geographic location and policies of kidney biopsies from 2-10% in different studies [3-13]. In our series it accounted for 2.9% which is similar to reported in Gupta R et al., [4]. CrGN is one of the most important pathologic correlate of rapidly progressive renal failure. It is classified into anti-GBM disease (linear IgG staining) (Type I), immune complex mediated (Type II) (granular glomerular staining on immunofluorescence), pauci-immune CrGN (TypeIII) (little or no staining for immunoglobulins or complement) and combination of anti-GBM & pauci-immune (Type IV) [1-3].
In our study, the most common aetiology for CrGN was Pauci-immune (41%) followed by immune complex GN (28.3%). Report from the university of North Carolina nephropathology laboratory showed Pauci-immune CrGN as the most common (60%) followed by immune complex disease (24%) and anti GBM(12%) in overall population [1]. Similarly, in Gupta R et al., pauci-immune was the most common group (71.7% of cases) followed by immune complex -mediated CrGN (28.3%) [4], whereas in Rampelli SK et al., from southern India, the most common aetiology found was Immune complex (IC) disease [7]. Reports from China and Saudi Arabia have also shown a relatively higher incidence of IC-mediated CrGN [3,10,11,13]. This has been attributed probably to the high prevalence of infection with nephritogenic strains and regional and epidemiological variations.
In the present study, Anti-GBM disease (24%) was more prevalent compared to other previously concluded Indian studies [4,7]. The pauci-immune CrGN had only 50% ANCA positivity in our study. This may be due to variation in socioeconomic status and study population.
Similar to Rampelli SK et al., the predominant immune complex disease in our population was IgA nephropathy [7]. The patients with immune complex disease had a better outcome compared to Anti-GBM disease and pauci-immune CrGN [Table/Fig-5] in our study and is similar to described in existing literature [1].
Final outcome based on aetiology of CrGN.
| CrGN type | Final Outcome (N=27*) | Total | p-value |
|---|
| Complete/ partial recovery | End stage Renal disease/ Mortality | N=27* | |
|---|
| Anti GBM | 2 | 5 | 7 | 0.03 |
| Immune Complex | 6 | 1 | 7 |
| Pauci immune | 2 | 9 | 11 |
| Anti GBM+ ANCA | | 1 | 1 |
| Others (TMA with Malignant Hypertension | 0 | 1 | 1 |
* Out of 29 studied, 2 patients lost to follow-up (one in Immune complex and one in Pauci-immune group).
The prognosis in CrGN is dependent on the age, aetiology, extent of the renal failure and the histological subtype [1-13]. A strong predictor of outcome for all types of CrGN is the severity of renal insufficiency at the time of presentation [1]. The other unfavorable predictors are elderly patients, presence of oliguria, requirement of haemodialysis, very late presentation, >75% circumferential crescents, Fibrous crescents and IFTA in histopathology [1-13].
The overall prognosis in our study remained poor with 30% reaching ESRD and 27.6% mortality. It is similar to Rampelli SK et al., who reported 48.6% of ESRD and 18.9% of mortality [7].
High creatinine at presentation (>7.2 mg/dl) and more anti GBM and ANCA negativity in pauciimmune CrGN patients in the study group may be the reason for this bad outcome in our study. Few other Indian studies have reported low rates of ESRD and mortality in contrast to our study [5,6,12]. In those studies, patients had a much lower serum creatinine levels at presentation and included children. In our study, the significant predictors of poor outcome on univariate analysis were oliguria at presentation (p=0.02), serum creatinine at presentation (>5.5mg/dl) (p=0.003) and requirement of haemodialysis (p=0.001). It is similar to previously done studies in literature [1,5,9,13].
Among histopathological features, the percentage of glomeruli with fibrocellular/fibrous crescents and IFTA were higher in non-responders but on univariate analysis both did not have statistically significant influence {(fibrous/fibrocellular crescents (p-0.34) or IFTA (p-0.23)} on the outcome in our study. In Gupta R et al., the only factor in histopathology which predicted a poor outcome was arteriolar fibrinoid necrosis [4]. In our study, no patients had arteriolar fibrinoid necrosis.
Limitation
The limitations in our study were single centre, small study population, short duration of follow-up and retrospective in nature.
Conclusion
In our cohort, the most common cause for diffuse CrGN is pauci-immune CrGN. Diffuse CrGN carries a poorer prognosis. Patients with pauci-immune and Anti-GBM disease have worse prognosis compared to immune complex CrGN. The presence of oliguria, high serum creatinine and requirement of haemodialysis at admission are associated with poor outcome.
*Data represented in Median (I R- Inter-quartile range), †- %- Percentage, ‡ (mean ± SD), SCr-serum creatinine
**Data represented in Median (IR- Inter-quartile range), †- %- Percentage, IFTA-Interstitial fibrosis tubular atrophy, ATN-Acute tubular necrosis, SCr-serum creatinine
* Out of 29 studied, 2 patients lost to follow-up (one in Immune complex and one in Pauci-immune group).