Azithromycin is one of the broad spectrum antibiotic and most commonly prescribed macrolide for upper respiratory and lower respiratory tract infections on an outpatient basis [1]. Among the macrolide antibiotics, azithromycin has minimal proarrhythmic activity as its side effect, yet retrospective analysis on five days of azithromycin is reported to have increased cardiovascular death compared to amoxicillin and ciprofloxacin. Estimated incidence was 47 deaths per 1 million cases [2].
Based upon this in 2013, FDA issued a black box warning regarding azithromycin usage as it may induce a prolongation of QTc resulting in torsade’s de pointes; hence, clinicians should consider risk benefit score before prescribing it [3].
Antihistamines like cetirizine even though known to cause prolongation of QT interval comes under low risk category and are commonly prescribed for upper respiratory infections along with azithromycin [4,5]. However, in recent past there is a gap in knowledge about the conflicting views on azithromycin and its cardiovascular morbidity and there is no study which evaluated the effect of azithromycin and cetirizine when prescribed for upper respiratory tract infections [1].
The purpose of the study was to assess the effect on QTc among patients treated with azithromycin and cetirizine combination for upper respiratory tract infection and objective of the study was to compare the baseline QTc with day 4 QTc, to see the proportion of patients in whom there is a significant change in QTc and to see the proportion of patients in whom QTc is crossing upper limit of normal on day 4 of ECG.
Materials and Methods
We conducted a clinical follow up (longitudinal) study at teaching hospital affiliated to Kasturba Medical College, Manipal University, Mangaluru, Karnataka, India. A total of 45 patients who were prescribed azithromycin and cetirizine for upper respiratory tract infections on outpatient basis; were studied from October 2014 to July 2016. All these patients were followed for a period of four days. Sample size was calculated based on the formula, n = (Zα)2σ2÷d2.
n = sample size; Zα = 1.96 (value for attaining 5% significance); σ = population standard deviation; d = minimum clinically important difference we are willing to detect.
By using this formula, with 95% confidence interval and 80% power of the study, sample size was calculated as 45. The approval for conducting the study was taken from the the Institutional human Ethical Committee, Kasturba Medical College, Manipal University, (Reg. No. ECR/541/Inst/KA/2014). Written informed consent was obtained from all the patients in their own language.
Inclusion criteria: All patients (>18 years) who received azithromycin and cetirizine for three days for upper respiratory tract infections.
Exclusion criteria:
Patients with baseline ECG showing QTc interval above upper limit.
Patients on drugs that is known to cause QT prolongation [4].
Critically ill patients.
Patients already on azithromycin for infections other than upper respiratory tract infection.
After explaining the details about the study, an informed consent was taken from the concerned patient. ECG was taken on day 1 before starting azithromycin and cetirizine and baseline QTc was calculated as described below. All the subjects were followed up for four days. During the follow up of patients on day 4, repeat ECG was taken and QTc was calculated.
QT interval is a measure of distance between the beginnings of QRS complex to the end of T wave on a 12 lead ECG recorded at 25 mm/sec at amplitude of 0.5 mV/cm. The point of intercept between the tangent line drawn to the descending portion of the T wave and isoelectric line was taken as the definite end of T wave and Lead II was used to measure it [Table/Fig-1] [6,7].
Measurement of QT interval.
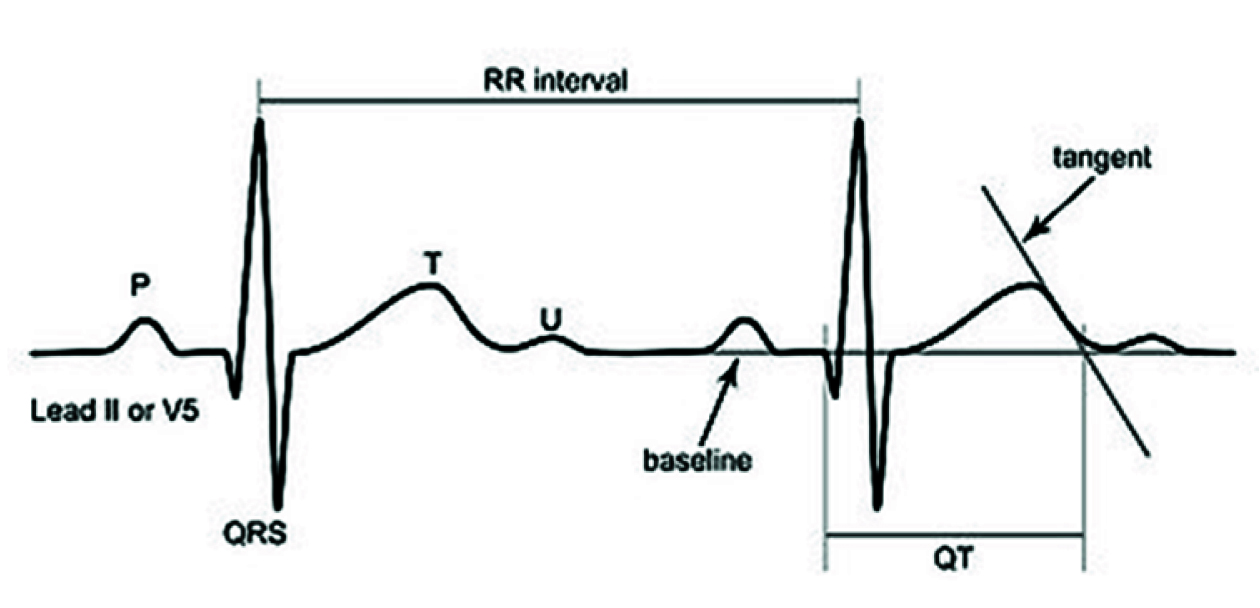
QT was corrected for an individual’s heart rate using Bazette’s formula, QTc = QT/RR [7]. RR interval is the distance between two consecutive R wave in an ECG. Normal value of QTc is between 380-430 ms.
Statistical Analysis
Statistical analysis was done comparing day 1 and day 4 QTc using students paired t-test.
Results
A total of 45 URIs patients on azithromycin and cetirizine who fullfilled the criteria were studied and results were analysed.
The statistical analysis of above obtained data revealed that the change in QTc on day 4 of ECG is highly significant with the p-value <0.001 [Table/Fig-2].
Comparison of day 1 QTc and day 4 QTc.
| Statistics | Day 1 QTc | Day 4 QTc |
|---|
| No. of patients | 45 | 45 |
| Mean | 388.80 ± 20.071 | 408.13 ± 24.101 |
| t-value | 8.723 |
| p-value | <0.001 |
In 43 patients out of 45, there is a change in QTc on day 4 compared to day 1 of ECG (95.6%). In two patients, QTc remains the same.
On further analysis, the day 4 QTc crossed above normal limit in only 8 (17.8%) subjects and using binomial test, this was not statistically significant. Among the subgroup of 37 patients in whom the day 4 QTc was within normal limit, the number of patients in whom there was change in QTc was statistically significant with a p-value <0.001.
Discussion
First prospective observational study to know the effect of QT interval with azithromycin in healthy people done by Strle F and Maraspin V including 31 female and 16 male participants of age 19-77 years [8]. Participants received 500 mg of azithromycin twice for the first day, followed by 500 mg once a day for the rest four days.
Analysis of ECG that was done; before initiating azithromycin, on 7th day and 14th day showed QTc of 406 ms, 412.5 ms and 419 ms respectively. However, proportion of patients in whom QTc was found to be above the upper limit of normal (440 ms) remained same before and after treatment [8].
In our study, participants between ages of 20-68 years who were treated with azithromycin and cetirizine had a QTc of 388.8 and 408.13 on 1st day and 4th day respectively, which is consistent with the above mentioned study.
A retrospective cohort study done in U.S Medicaid benefaciaries by Ray WA et al., revealed that azithromycin had significant risk of cardiovascular mortality which was 2-3 times higher when compared to amoxycillin or without any antibiotic in patients with pre-existing cardiovascular morbidity [2].
Above study led to FDA issuing a black box warning regarding usage of azithromycin for patients with cardiovascular morbidity.
Another retrospective study done by Svanstrom H et al., on Danish population suggested no increase in cardiovascular mortality with azithromycin in comparision to penicillin V [1].
Since, above mentioned studies gave conflicting views, regarding risk of cardiovascular mortality with azithromycin, this has remained controversial and requires further studies. Main limitations of these studies being that they were done in retrospective manner, baseline cardiovascular risk was not matched and moreover indication for use of azithromycin was not clearly defined.
Our study eliminates the above mentioned limitations and is a prospective study with respect to use of azithromycin for upper respiratory tract infections.
Our study analysed the effect of a commonly prescribed combination of azithromycin and cetirizine on QTc interval in patients with upper respiratory tract infection and found that change in QTc occurred in 96.4% of patients.
Among our study population, 8 out of 45 patients had an increase in QTc interval above the upper limit of normal (430 ms) making them more susceptible to cardiac arrhythmias and may need more follow up.
Long term follow up of patients was not possible, especially for patients in whom QTc is above 430 ms on day 4. A larger sample size may be needed to study the group of patients in whom the QTc goes above 430 ms on day 4.
Strength of The Study
Our study is the first prospective study to analyse the risk of azithromycin and cetirizine combination, which is frequently used for upper respiratory tract infections.
This study was done on outpatient basis on healthy individuals who were not on any other drugs eliminating the effect of drug interaction.
This study being prospective is done in patients with normal baseline QTc thus eliminating bias found in retrospective analysis (congenital long QTc, baseline electrolyte abnormality or cardiovascular risk).
Limitation
Long term follow up of patients was not possible, especially for patients in whom QTc was above 430 ms on Day 4. Whether the prolonged QTc actually resulted in any significant arrythmias were not a part of our study, which would be clinically important. A larger sample size may be needed to study the group of patients in whom the QTc was prolonged. The duration for which the QTc remained prolonged was also not studied.
Conclusion
Clinicians need to be aware that treatment of a simple URIs with a three day course of azithromycin and cetirizine can cause significant increase in the QT interval and in certain instances increase it beyond the upper limit of normal as well. Hence, patients should be followed on day 4 with ECG. A closer follow up will be required in patients with QTc prolongation (>430 ms).