Case Report
A 27-year-old male, farmer by occupation, resident of village located nearby Sangamner, Maharashtra, presented with complains of foreign body sensation, redness, itching, discharge from the right eye since five days.
On asking details, he gave history of injury to cornea of right eye by sudden accidental inoculation of small insects (size less than mosquito as described by patient), following which he tried to remove it with fingers. From next day patient started complaining of redness, itching and discharge from right eye and developed white patch on cornea which was progressed gradually to size of diameter of pea. Patient also complained about blurring of vision of right eye and low grade fever. No history of topical application of corticosteroids or herbal juice was noted. Patient was non diabetic and non hypertensive.
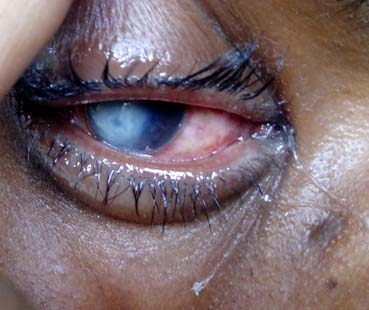
On local examination of right eye, cornea showed ulcer approximate 6-7 mm in size, irregular in shape, white opaque in colour with irregular margin [Table/Fig-1]. Vision was impaired in right eye. No abnormality was detected in left eye. Routine blood examination was within normal limit. Clinical case was diagnosed as corneal ulcer and patient was put on local symptomatic and antibiotic treatment like atropine, ibuprofen plus paracetamol, amoxicillin- clavulanic acid and topical antibiotic eye drops like ciprofloxacin (0.3%), moxifloxacin (0.5%) to which patient did not respond. Hence, it was decided to do an investigation to rule out fungal infection.
Corneal ulcer 6-7 mm in size, irregular in shape, white opaque in colour with irregular margin.
Microbiological Examination
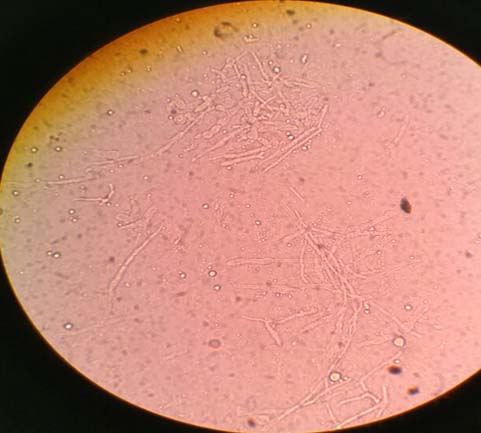
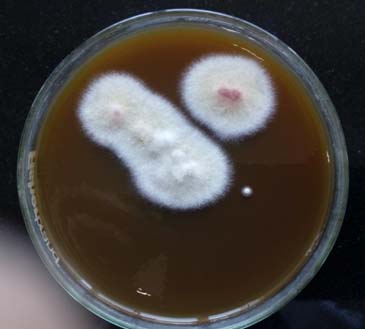
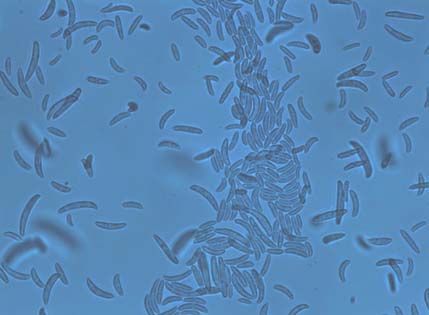
Scraping from the cornea was collected using Kimura spatula with all aseptic precautions. Scraped material was processed for KOH wet preparation and Gram staining. KOH wet preparation revealed fragments of septate, branched, hyaline hyphae with swelling at some points [Table/Fig-2]. Gram smear examination revealed few pus cells and few Gram positive fragments of septate hyphae. Further, the sample was processed for bacterial culture (on MacConkey’s agar and blood agar) and fungal culture on Sabouraud Dextrose Agar (SDA). MacConkey and blood agar were incubated at 37°C for 24 hours. SDA was incubated in two sets at two different temperatures (30 and 37°C) for three weeks. No bacterial growth was observed on MacConkey and blood agar within 24 hours although fungal colonies were grown on blood agar within four days. On SDA also, fungal colonies were visible within four days. The colonies were wooly, cottony, flat and white coloured at first which then produced aerial mycelium with pinkish pigment in center of the colony [Table/Fig-3]. Lactophenol Cotton Blue (LPCB) mount revealed hyaline branched septate hyphae, long monophialidic conidiogenous cells, moderately curved macroconidia and oval microconidia. Slide culture was done to identify the species which showed three to five septate, fusiform, cylindrical, often moderately curved macroconidia, with an indistinctly pedicellate foot cell [Table/Fig-4]. Microconidia were wider, cylindrical to oval, with thicker wall and were single-celled to two-celled [Table/Fig-5,6]. The isolated colony was also subjected to urease test which came as negative. Slide culture from the colonies grown on Potato Dextrose Agar (PDA) revealed chlamydospore production. They were produced singly, oval in shape and smooth walled [Table/Fig-7]. Therefore, on the basis of KOH mount, colony morphology, colour of pigment produced, microscopic picture on LPCB (morphologic characteristics of macroconidia, microconidia, presence of long monophialidic conidiogenous cells, production of chlamydospores), urease test and slide culture, it was identified as Fusarium solani. The ophthalmologist was informed the report and patient’s treatment was switched from antibiotics to natamycin (5% eye drops two hourly for one month). In the follow up OPD visit of the patient after two weeks, signs and symptoms were found to be improved.
KOH wet preparation microscopy (at 400x) showing branched, septate, hyaline hyphae of Fusarium solani.
Colony of Fusarium solani on SDA.
Lacto-Phenol Cotton Blue microscopy (at 400x) showing three to five septate, fusiform, cylindrical often moderately curved macroconidia.
Long monophialidic conidiogenous cells of Fusarium solani.
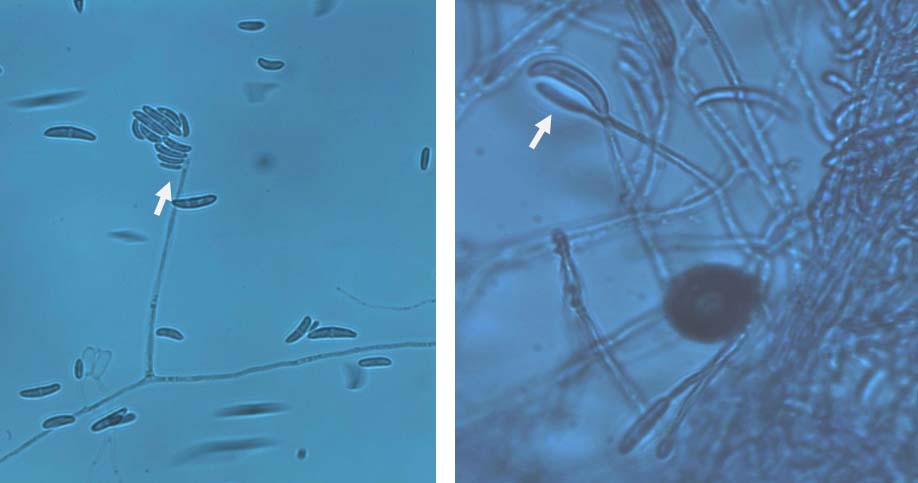
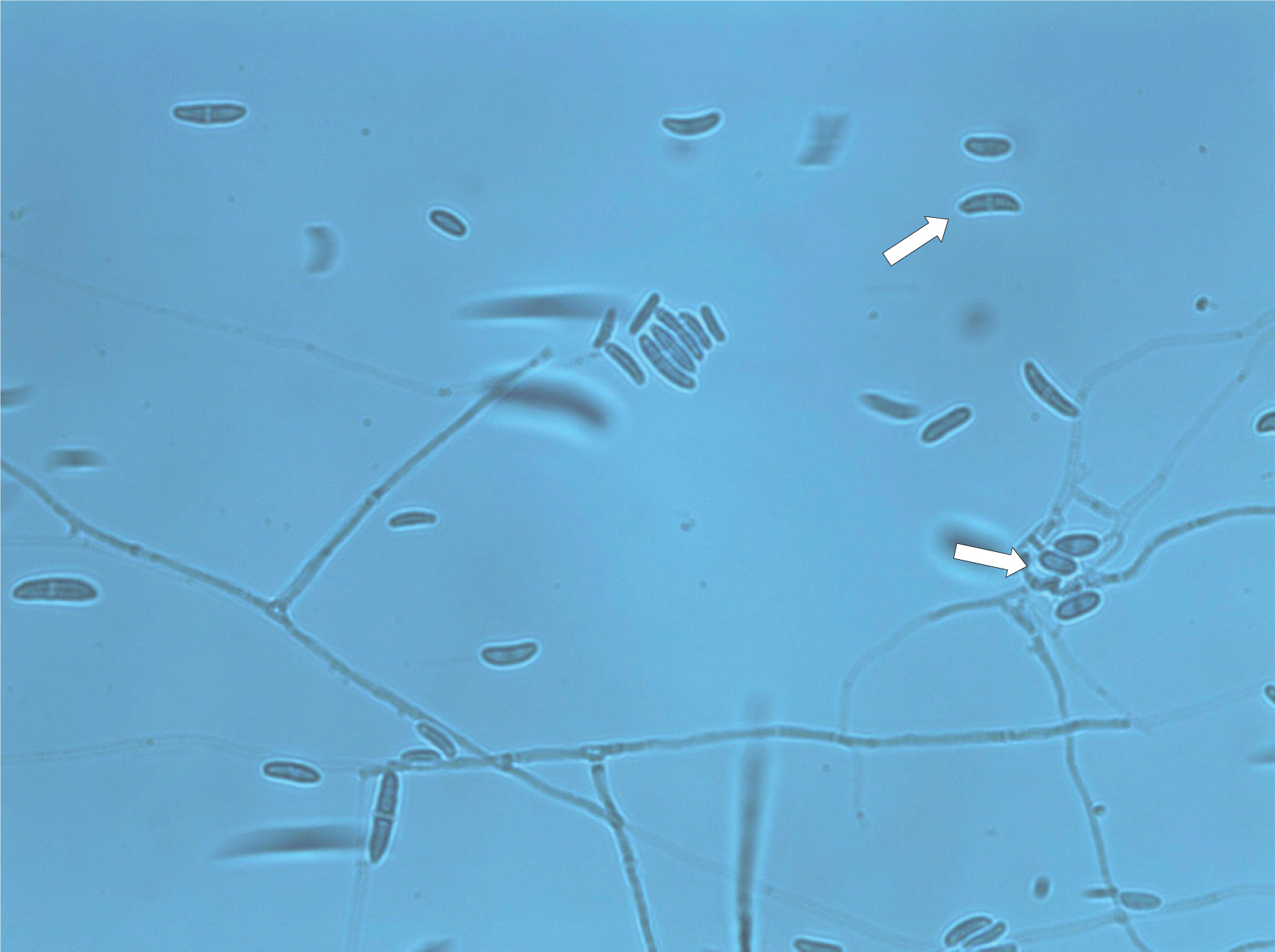
One or two celled, oval microconidia of Fusarium solani (shown with arrows).
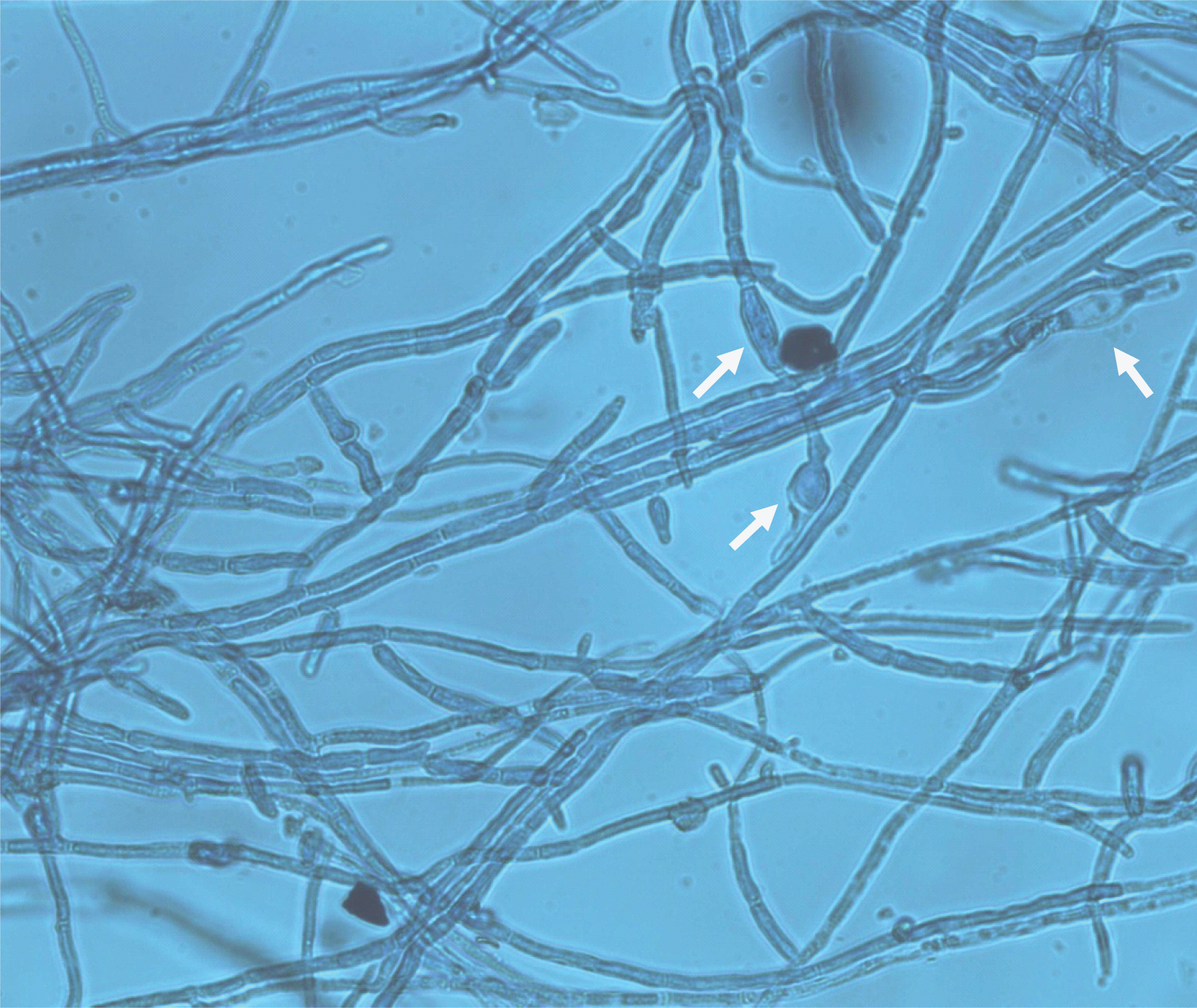
Single, oval and smooth walled chlamydospore produced by isolated strain of Fusarium on PDA (shown with arrows).
Discussion
The causative fungal species of keratomycosis are ubiquitous organisms, which are responsible for 6-53% of all corneal infection globally. These kinds of infections are frequently encountered resulting visual disability, especially in tropical countries. Corneal opacification following different kinds infections is second most common cause of blindness after cataract [1].
The filamentous fungi are responsible for up to one third of all cases of keratitis in certain parts of the world. Aspergillus and Fusarium are responsible for one third of all traumatic keratitis. Other fungal genera which also can cause infections include, Curvularia, Candida, Acremonium, Paecilomyces, Penicillium, Alternaria, Fonsecaea, Pseudallescheria, Bipolaris, Aureobasidium [1,2].
Due to large rural population and environmental factors, keratomycosis is common in India. Injury to the cornea is the leading cause of microbial keratitis, particularly fungal keratitis. The nature of injury is often vegetative in origin, which may consist of trauma with plant twigs, rice-husk, cotton plant, etc., [3]. A history of trauma to cornea with organic or vegetable matter is reported in 55%–65% cases of fungal keratitis [3]. Trauma to the cornea causes destruction of the epithelium and Bowman’s membrane, which acts as barrier for infection. The stroma becomes overhydrated and is altered in such a way that it becomes a more favourable site for fungus to grow. Keratomycosis is an occupational hazard of agricultural workers. The seasonal variation observed in other studies may be due to occupational injuries occurred during harvesting [4]. This finding matches with our case report where, patient is farmer by occupation and also gave history of corneal trauma by accidental inoculation of insect in his right eye, which he tried to remove it with his fingers. Although some of the observers have not reported the history of corneal trauma in their cases [5,6]. Also, Fusarium solani may serve as source of food for some tree-boring insects as described by Hara H et al., [7]. The patient is also from rural area and this incident occurred during harvesting season only.
Fusarium species have also been reported from patients suffering from endophthalmitis, otitis media, onychomychosis, cutaneous infections particularly of burn wounds, breast abscess mycetoma, sinusitis, pulmonary infections, endocarditis, peritonitis, central venous catheter infections, septic arthritis, fungemia [8,9]. Clusters of Fusarium keratitis were also reported among contact lense user [10].
Laboratory diagnosis is crucial in the management of mycotic keratitis. Direct demonstration of the fungus in the smear leads to an immediate diagnosis. Examination with 10% KOH wet mount has been proved extremely useful with over 86% positivity in some of the studies [11]. The true hyphal fragments can usually be recognized under low power and confirmed by higher magnification. Gram stain will also reveal fungal filaments. In our case, we have found septate, branched, hyaline hyphae abundantly with swelling at some points on KOH and Gram positive filaments on Gram stain.
Fusarium solani grows rapidly at 37°C unlike most fungi which grow better at 30°C. It grows well on media used for bacterial culture. On SDA, colonies were wooly, cottony and white coloured at first which then became fluffy pink coloured. LPCB mount revealed hyaline branched septate hyphae, long monophialidic conidiogenous cells which differentiates Fusarium solani from Fusarium oxysporum. LPCB from slide culture method showed three to five septate, fusiform, cylindrical, often moderately curved macroconidia, with an indistinctly pedicellate foot cell. Microconidia were wider, cylindrical to oval, with thicker wall. The isolate did not produce urease which is also a differentiating feature of Fusarium solani from Fusariumoxysporum. Slide culture from the colonies grown on Potato Dextrose Agar (PDA) revealed chlamydospore which was produced singly, oval in shape and smooth walled. Our observation also matches the findings described by Anandi V et al., and Ismail MA et al., [9,12].
The advent of antibiotics and steroids has been blamed for the increasing incidence of fungal keratitis [13]. In our case, after reporting the fungal species, antibiotics were withdrawn and patient was started on natamycin. The signs and symptoms were improved after start of the antifungal therapy.
Conclusion
We report the uncommon Fusarium solani isolated from a case of keratomycosis. Most important laboratory investigation of fungal corneal ulcer is direct microscopic examination and culture which are simple and does not require costlier set up. An early specific diagnosis is essential for initiation of early antifungal therapy to avoid inappropriate treatment of antibiotics and corticosteroids which instead of curing only worsen the disease.
[1]. Chander J, Miscellaneous mycoses, related topicsIn: Textbook of Medical Mycology 2012 3rd edNew DelhiMehta Publishers:400-09. [Google Scholar]
[2]. Vajpayee RB, Gupta SK, Bareja U, Kishore K, Ocular atopy and mycotic keratitisAnn Ophthalmol 1990 22:369-72. [Google Scholar]
[3]. Bharathi MJ, Ramakrishnan R, Vasu S, Meenakshi R, Palaniappan R, Epidemiological characteristics and laboratory diagnosis of fungal keratitis: a three-year studyIndian J Ophthalmol 2003 51:315-21. [Google Scholar]
[4]. Ahmed I, Owais M, Shahid M, Aqil F, Combating fungal infectionsProblems and Remedy. Berlin 2010 :77-78. [Google Scholar]
[5]. Hamdan JS, Resende MA, Franzot SP, Dias DV, Viana EM, Casali AK, A Case of mycotic keratitis caused by Fusarium solaniRev Inst Med trop Sao Paulo 1995 37(2):181-83. [Google Scholar]
[6]. Vijaya D, Sumathi Malini, Keratomycosis due to Fusarium oxysporum- a case reportIndian J Pathol Microbiol 2001 44(3):337-38. [Google Scholar]
[7]. Hara H, Beardsley JW, The biology of black twig borer Xylosandrus compactus in Hawaii, USAPreceedings Hawaiian Entomol Soc 1979 23:55-70. [Google Scholar]
[8]. Prajna L, Vijaykumar Prajna NV, Srinivasan Aravind’s atlas of fungal corneal ulcersClinical Features & Laboratory Identification Methods 2008 1st edNew DelhiJaypee Brothers Medical Publishers:78-79. [Google Scholar]
[9]. Anandi V, Vishwanathan P, Sasikala S, Rangarajan M, Subramaniyan CS, Chidambaram N, Case Report Fusarium solani breast abscessInd J Med Microbiol 2005 23(3):198-99. [Google Scholar]
[10]. Guarro J, Rubio C, Gene J, Cano J, Gil J, Benito R, Case of keratitis caused by an uncommon fusarium speciesJ Clin Microbiol 2003 41(12):5823-26. [Google Scholar]
[11]. Sharma S, Srinivasan M, George C, The current status of fusarium species in mycotic keratitis in south IndiaIndian J Med Microbiol 1993 11(2):140-47. [Google Scholar]
[12]. Ismail MA, Abdel-Hafez SAA, Hussein NA, Hameed NAA, Contributions to the genus fusarium in Egypt with dichotomous keys for identification of species 2015 1st edPolandTomasz M. Karpinski:88-89. [Google Scholar]
[13]. Singh H, Jamal F, Marahakim Song CC, Fusarium solani keratitis first report from MalasiaMed J Malasia 1981 36(2):89-91. [Google Scholar]