Cauda Equina Syndrome in a Lactating Mother - A Safe Treatment Approach
Ajay Kothari1, Ketan Khurjekar2, Shailesh Hadgaonkar3, Himanshu Kulkarni4, Parag Sancheti5
1 Assistant Professor, Department of Spine Surgery, Sancheti Institute of Orthopaedics and Rehabilitation, Pune, Maharashtra, India.
2 Chief Consultant, Department of Spine Surgery, Sancheti Institute of Orthopaedics and Rehabilitation, Pune, Maharashtra, India.
3 Deputy Chief Consultant, Department of Spine Surgery, Sancheti Institute of Orthopaedics and Rehabilitation, Pune, Maharashtra, India.
4 Junior Consultant, Department of Spine Surgery, Sancheti Institute of Orthopaedics and Rehabilitation, Pune, Maharashtra, India.
5 Director, Department of Orthopaedics, Arthroscopy and Arthroplasty, Sancheti Institute of Orthopaedics and Rehabilitation, Pune, Maharashtra, India.
NAME, ADDRESS, E-MAIL ID OF THE CORRESPONDING AUTHOR: Dr. Himanshu Kulkarni, 11/12 Thube Park, 16, Shivajinagar, Pune-411005, Maharashtra, India.
E-mail: hgkulkarni@gmail.com
Cauda equina syndrome is widely considered as a surgical emergency. The cause of cauda equina syndrome usually is a large central lumbar disc herniation, prolapse or sequestration. Decompression at the earliest has been suggested by many authors but the planning of surgical management becomes challenging when the patient is a breastfeeding mother. Fear of harmful effects of the drugs (administered in the mother) on the infant, always confuses clinicians regarding the treatment approach. So the multidisciplinary approach is necessary with involvement of anaesthetist, paediatrician and also a gynaecologist if necessary. Thorough knowledge of the safety of drugs to be used in operative and post operative period becomes a necessity keeping the baby into consideration. We present a case of one month postpartum female with cauda equina syndrome and present a stepwise multidisciplinary approach, which involves active contributions from surgeon, for safety of the mother and the infant.
Incomplete cauda equina syndrome, Pregnancy related low back pain, Propofol
CASE REPORT
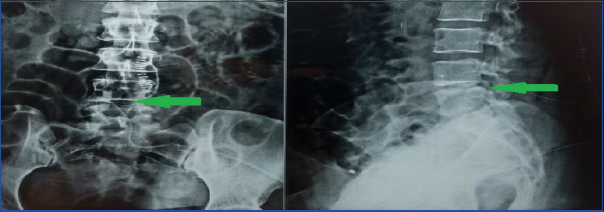
A 27-year-old lady came to casualty with weakness in both lower limbs since one day. She also had a complaint of difficulty while micturition in form of having a poor stream and more strain needed to micturate. Patient had delivered a baby one month back by normal vaginal delivery, but was suffering from back pain since then. The back pain was also aggravated since one day. Patient didn’t have any history of trauma or fall or strenuous activity. Neurological examination of the patient revealed paraparesis. Patient was found to have grade 0 power for both ankle and great toe dorsiflexion and plantar flexion. Patient had typical saddle anaesthesia but trigone sensations were preserved which was checked with tugging of a Foley’s catheter. X-ray of the patient showed reduced disc space at L4-L5 [Table/Fig-1] but no obvious bony abnormality. Magnetic Resonance Imaging (MRI) scan of the lumbar spine was performed. MRI revealed a large posterocentral and right paracentral disc extrusion [Table/Fig-2] at L4-L5 level with inferior migration of the disc. The extrusion of disc measured 1.9 cm x 1.2 cm in dimension, causing severe canal stenosis, thecal sac and traversing root compression with complete loss of signal on MR myelogram [Table/Fig-3].
Large extruded disc at L4-L5 with inferior migration.
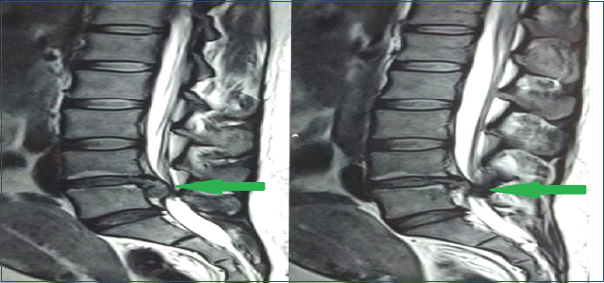
Large posterocentral and right paracentral disc extrusion.
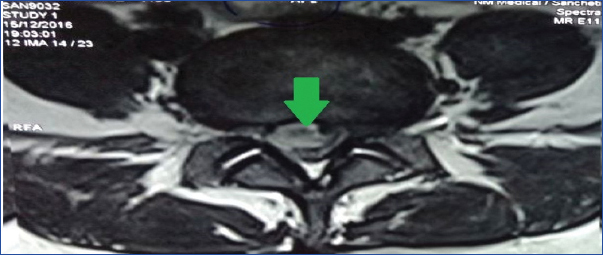
Complete loss of signal on MR myelogram.
In view of clinical and radiological picture, the patient was diagnosed to have Incomplete Cauda Equina Syndrome (CESI). Since patient was a breastfeeding mother, preoperative planning was done meticulously. Opinion of the paediatrician and the anaesthetist was taken, breastfeeding was done one hour before surgery and mother’s milk was stored by breast pumping just before surgery. For induction of anaesthesia, ultra short acting anaesthetic drugs in form of propofol and fentanyl were used. A short acting safe inhalational agent in the form of sevoflurane was used. Patient was given prone position and level was marked. Midline incision was taken and laminectomy was performed. Thorough decompression was done and L4-L5 disc was removed [Table/Fig-4].
Postoperative X-ray showing L4-L5 decompression.
Level was confirmed again. Exiting and traversing roots were made completely free. Local infiltration of skin was done with bupivacaine instead of lignocaine for postoperative analgesia and suturing was done over a drain. Breastfeeding wasn’t started until 24 hours of surgery. Intravenous drips of paracetamol were used for analgesia for 24 hours and then ibuprofen was given orally. Amoxicillin with clavulunate was used as an intravenous antibiotic for five days and then same drug was given orally for three days according to Hospital antibiotic policy. Postoperative physiotherapy was started after 24 hours. Patient improved progressively after four month of follow up. Saddle anaesthesia, motor weakness and urinary complaints of the patient were completely reverted and patient also regained Grade 4 power at both ankles.
Discussion
Cauda equina syndrome (CES) is widely considered as a surgical emergency. The cause of CES usually is a large central lumbar disc herniation, prolapse or sequestration. The incidence of CES is considered to be 0.07% to 0.12% amongst all prolapsed lumbar intervertebral discs [1,2]. Decompression at the earliest has been suggested by many authors but the planning of surgical management becomes challenging when the patient is a breastfeeding mother. With the advancement in field of anaesthesia and pharmacology, there are several management options that are available but are underutilized because of the fear of harmful effects on the baby via breast milk. So in these cases, multidisciplinary approach is necessary with involvement of anaesthetist, paediatrician and also a gynaecologist if necessary. Thorough knowledge of the safety of drugs to be used in operative and postoperative period becomes a necessity keeping the baby into consideration. So we have put forth the do’s and don’ts for safety of mother and the baby from perspectives of different groups in the treating team.
From a Spine surgeon’s point of view, CES in a second postpartum month is a rare scenario. Though Pregnancy Related Low Back Pain (PLBP) and Pelvic Girdle Pain (PGP) are very common (approximately 25% of all postpartum women) [3], progression of PLBP to CES is very rare. Pathophysiology of a PLBP is related to relaxin that is produced in increased quantities by both corpus luteum and the uterine decidua during pregnancy [4]. It leads to relaxation of the connective tissue which further leads to greater ligament laxity, particularly in sacroiliac joint [5]. Although it’s the most common cause of back pain in postpartum period, lumbar disc herniation, though uncommon during and after pregnancy, should always be excluded as it has been found on MRI, finding of lumbar disc herniation in pregnant females with back pain has the same prevalence as in non pregnant females [6,7]. Progression of these disc prolapses labelled as PLBP can land into catastrophic neurological complications in form of CES.
CES can be roughly divided into two types, complete or Incomplete, in relation to urinary function and perineal sensation. In CESI, the urinary difficulties of the patient are of neurogenic origin. These are, altered urinary sensation, poor urinary stream, loss of desire to void and the need to strain in order to micturate. Patient also has unilateral or partial saddle and genital sensory deficit with trigone sensation usually intact. The Cauda Equina Syndrome Retention (CESR) or complete syndrome has painless urinary retention and overflow incontinence. This type of CES has complete saddle and genital sensory deficit and trigone sensation is usually lost [8]. One way to differentiate between the two is trigone sensation. An inflated Foley’s catheter (inserted into the bladder) is gently pulled when the patient is unaware. Pulling of the catheter produces the micturition urge and this will help to distinguish the patients with pain-related retention from the patients with genuine neurological deficit [9].
Timing surgery for CES has been most widely debated. Although many authors have stated that surgery at the earliest (within 24-48 hours) [10,11] gives best results, some state that there is no evidence to support the view that emergency surgery influences the degree of recovery [12,13].
From an anaesthetist’s point of view, whatever the timing of surgery is, carrying it out in a breastfeeding mother is always challenging considering the effects of drugs passed to the baby via mother’s milk. The crux of the anaesthetic drugs to be used in these patients is that the drugs have to be ultra short acting and have to be completely metabolised from mother’s body as soon as possible. The transfer of any medication into breast milk depends on pharmacological properties of the drug including protein binding, lipid solubility, molecular weight and maternal plasma level of the drug [14]. In general, medications that are highly lipid soluble, less protein bound and with lower molecular weight are associated with greater penetration into breast milk. Anaesthetic agents could potentially cause problems in newborn infants but they have minimal to negligible effect on older infants. So mothers with older infants can generally resume breastfeeding as soon as they are awake, stable and alert [15]. Resumption of normal feeding is the indication that these medications have been redistributed from the plasma compartment (which is the milk compartment) and have entered adipose and muscle tissue. Drugs which are highly lipid soluble can sometimes be an exception as the breast tissue may function as a fat compartment, which will be a drug reservoir. For anaesthesia in obstetric patients, propofol is considered to be an ideal agent for induction, taking its short duration of action, antiemetic profile, rapid metabolism with no active metabolites and its negligible milk levels after two hours into consideration. Breastfeeding can be started in immediate postoperative period and it is safe, after administration of propofol, due to it’s low bioavailability and rapid metabolism [16]. Among other drugs for induction, etomidate and midazolam are also considered safe with rapid clearance of drug and negligible milk concentration after 24 hours [17,18]. Inhalational anaesthesia agents like sevofurane, isoflurane, halothane are rapidly excreted and have poor bioavailability. Hence, they are considered safe and don’t have any hazardous effects on the baby via milk [15]. But amongst all, sevoflurane is considered to be the safest for the patient, hence it is preferred over others. Amongst the opioid derivatives, fentanyl is the safest one with low bioavailability and less that 0.1% of the administered drug present is in milk after 24 hours. By composition, neostigmine and glycopyrrolate are quaternary ammonium compounds. Since quaternary ammonium compounds don’t cross blood milk barrier, they are considered safe for reversal of anaesthesia [19]. For immediate postoperative analgesia, local anaesthetic injection in subcutaneous area is found to be safe and effective since systemic uptake from subcutaneous area is negligible. Bupivacaine is preferred over lignocaine in this matter for its rapid clearance [20].
From a paediatricians point of view, analgesics and antibiotics in the postoperative period is a key factor that a common clinician is most concerned about. For analgesia, paracetamol is most commonly used drug as it’s very safe for the baby. Ibuprofen is considered an ideal, moderately effective analgesic. Its transfer to milk is negligible with half-life of approximately 1.8 hours. So feeding can be resumed within 2-3 hours [21]. Ketorolac is also an excellent option as it gives better analgesia than ibuprofen and paracetamol and has extremely low levels in mother’s milk [22]. Celecoxib’s transfer into milk is extraordinarily low (< 0.3% of the weight adjusted maternal dose). Its short-term use is safe [23]. Opioid analgesics like morphine, codeine, meperedine should be avoided due to high levels in mothers milk which may cause problems like respiratory depression in infants and toddlers [24,25,26]. In postoperative period, beta lactam class of antibiotics are considered to be the safest amongst all [27]. They have minimal effusion into breast milk and have no harmful effects on the infants. So penicillins and cephalosporins, especially most established cephalosporins can be safely used in lactating period. According to German classification, use of aminoglycosides is restricted to very specific and very vital indications. Use of drugs like gentamycin and amikacin is contraindicated otherwise. Amongst macrolids, erythromycin and azithromycin are considered very safe [28]. They have both been considered indicated according to German classification and FDI guidelines. Other safe antibiotics that can be used during lactation period are vancomycin, rifampicin and isoniozid. Fluoroquinolones, which are another group of widely used potent antibiotics, are contraindicated in lactation period according to German Classification [28].
Conclusion
Though rare, CES in pregnancy is a possibility. The clinical examination should be thorough and intervention should be carried as soon as possible for a better outcome. Breastfeeding can be done till immediately before surgery, but should be ideally avoided for first 24 hours to allow all anaesthetic drugs to get metabolised from the body of mother so that they are not passed to the baby via milk. There are many anaesthetic drugs which are safe for anaesthesia in a lactating mother and specific analgesic and antibiotic drugs can also be administered postoperatively without hesitation. We have tried to put forth simple do’s and don’ts in case of drugs to guide clinicians for safe treatment approach.
[1]. Scorbie C, Cauda equina syndrome. Orthopaedics June 2009 - Volume 32 ·Issue 6 [Google Scholar]
[2]. Schoenfeld AJ, Bader JO, Cauda equina syndrome: an analysis of incidence rates and risk factors among a closed North American military populationClin Neurol Neurosurg 2012 114:947-50. [Google Scholar]
[3]. Wu WH, Pregnancy related pelvic girdle pain (PGP) I: terminology, clinical presentation, and prevalenceEur Spine J 2004 13:575-89. [Google Scholar]
[4]. MacLennan AH, Nicolson R, Green RC, Bath M, Serum relaxin and pelvic pain of pregnancyLancet 1986 2:243-45. [Google Scholar]
[5]. Brooke R, The sacroiliac jointJ Anat 1924 58:299-305. [Google Scholar]
[6]. Weinreb JC, Wolbarsht LB, Cohen JM, Brown CE, Maravilla KR, Prevalence of lumbosacral intervertebral disk abnormalities on MR images in pregnant and asymptomatic non pregnant womenRadiology 1989 170(Pt 1):125-28. [Google Scholar]
[7]. Alexander JT, McCormick PC, Pregnancy and discogenic disease of the spineNeurosurg Clin N Am 1993 4:153-59. [Google Scholar]
[8]. Gleave JRW, Macfarlane R, Cauda equina syndrome: what is the relationship between timing of surgery and outcome?Brit J Neurosurg 2002 16(4):325-28. [Google Scholar]
[9]. Uff CEClinical assessment of cauda equina syndrome and the bulbocavernosus reflex 2009 Accessed – March 31 2009See http://www.bmj.com/cgi/eletters/338/mar31_1/b396 [Google Scholar]
[10]. Ahn UM, Ahn NU, Buchowski JM, Garrett ES, Sieber AN, Kostuik JP, Cauda equina syndrome secondary to lumbar disc herniation: a meta-analysis of surgical outcomesSpine 2000 25(12):1515-22. [Google Scholar]
[11]. Kohles SS, Kohles DA, Karp AP, Erlich VM, Polissar NL, Time-dependent surgical outcomes following cauda equina syndrome diagnosis: Comments on a meta-analysisSpine 2004 29(11):1281-87. [Google Scholar]
[12]. McCarthy MJH, Aylott CRW, Grevitt MP, Bishop MC, Hegarty J, Cauda equina syndrome: factors affecting long term function and sphincteric outcomeSpine 2007 32(2):207-16. [Google Scholar]
[13]. Hussain SA, Gullan RW, Chitnavis BP, Cauda equina syndrome: outcome and implications for managementBrit J Neurosurg 2003 17(2):164-67. [Google Scholar]
[14]. Hale TW, Maternal medications during breastfeedingClin Obstet Gynecol 2004 47(3):696-711. [Google Scholar]
[15]. Montgomery A, Thomas WH, The Academy of Breastfeeding Medicine A, ABM Clinical Protocol #15:Analgesia and Anesthesia for the Breastfeeding Mother, Revised 2012 BREASTFEEDING MEDICINE Volume 7, Number 6, 2012 ªMary Ann Liebert, IncDOI:10.1089/bfm.2012.9977 [Google Scholar]
[16]. Dailland P, Intravenous propofol during cesarean section: placental transfer, concentrations in breast milk, and neonatal effects. A preliminary studyAnesthesiology 1989 71(6):827-34. [Google Scholar]
[17]. Esener Z, Thiopentone and etomidate concentrations in maternal and umbilical plasma, and in colostrumBr J Anaesth 1992 69(6):586-88. [Google Scholar]
[18]. Nitsun M, Pharmacokinetics of midazolam, propofol, and fentanyl transfer to human breast milkClin Pharmacol Ther 2006 79(6):549-57. [Google Scholar]
[19]. Dalal PG, Bosak J, Berlin C, Safety of the breastfeeding infant after maternal anesthesiaPaediatr Anaesth 2014 24(4):359-71. [Google Scholar]
[20]. Benjamin CobbBreastfeeding after anesthesia: A review for anesthesia providers regarding the transfer of medications into breast milkTransl Perioper Pain Med 2015 1(2):01-07. [Google Scholar]
[21]. Weibert RT, Townsend RJ, Kaiser DG, Lack of ibuprofensecretion into human milkClin Pharm 1982 1:457-58. [Google Scholar]
[22]. Drugs and lactation database (LactMed)U.S. National Library of Medicine, TOXNET Toxicology Data Networkaccessed October 18 2012http://toxnet.nlm.nih.gov/cgi-bin/sis/search/f?./temp/*bYSp38:1 [Google Scholar]
[23]. Hale TW, McDonald R, Boger J, Transfer of celecoxib into human milkJ Hum Lact 2004 20:397-403. [Google Scholar]
[24]. Halpern SH, Levine T, Wilson DB, Effect of labor analgesia on breastfeeding successBirth 1999 26:83-88. [Google Scholar]
[25]. Loubert C, Hinova A, Fernando R, Update on modern neuraxial analgesia in labour: A review of the literature of the last 5 yearsAnaesthesia 2011 66:191-212. [Google Scholar]
[26]. Hirose M, Hara Y, Hosokawa T, The effect of postoperative analgesia with continuous epidural bupivacaine after cesarean section on the amount of breast feeding and infant weight gainAnesth Analg 1996 82:1166-1169. [Google Scholar]
[27]. Czeizel AE, Rockenbauer M, Sorensen HT, Olsen J, Use of cephalosporins during pregnancy and in the presence of congenital abnormalities: a population-based, casecontrol studyAm J Obstet Gynecol 2001 184:1289-96. [Google Scholar]
[28]. Mylonas L, Antibiotic chemotherapy during pregnancy and lactation period: aspects for considerationArch Gynecol Obstet 2011 283:07-18. [Google Scholar]