Antimicrobial Resistance (AMR) continues to pose a significant problem for the healthcare industry both in Australia and abroad. AMR is developing rapidly-often within months of the release of new compounds and resistance incidence rates currently outstrip antimicrobial discovery and development [1]. Consequently, the number of antibiotic-resistant bacteria or ‘superbugs’, present in hospitals, nursing homes and the community continues to rise [2]. These superbugs are associated with increased patient morbidity and mortality, complexity and duration of treatment as well as increased hospital stay and consequently, substantial increases to healthcare system costs and financial burden to the community [1]. The strong association between antibiotic-prescribing practices in primary care and AMR rates indicates that GPs have an integral role in maintaining the efficacy of these essential drugs [2-4]. Key features of prescribing practice contributing to AMR include: inappropriate use (e.g., for viral upper respiratory tract infection; URTI), lack of compliance (missed doses, incomplete course, etc.,), and prolonged courses of both therapeutic and prophylactic treatment [1].
Australia is not immune to the international trend of increasing AMR. The resistance rate of Streptococcus pneumoniae, a common respiratory pathogen, to macrolide antibiotics has increased from 8.7% in 1994 to 20.4% in 2007 and further continues to rise [2]. Furthermore, since 2000, there has been a significant increase in Methicillin-Resistant Staphylococcus Aureus (MRSA), particularly in terms of community-acquired infection. In 2012, one in six patients with a staphylococcal infection were MRSA-positive and one in eight were infected with a community-acquired MRSA [5].
In 2012, in response to growing AMR and pressure from medical, veterinary, and public health sectors, the Australian Government established the Antimicrobial Resistance Standing Committee (AMRSC) to minimise AMR through surveillance, education and stewardship programs, infection prevention and control guidelines, and regulatory restrictions on the prescription and use of antimicrobials [1].
Given that most URTIs are viral in origin and are self-limiting, the prescription of antibiotics is frequently inappropriate [6]. Furthermore, systematic review concluded that antibiotics not only fail to reduce duration of illness or symptom severity, but increase morbidity due to adverse drug effects [7].
Given the ongoing relevance of antibiotic prescribing, this study aims to examine patient and clinician demographic factors, with respect to antibiotic prescribing patterns for URTIs in the community and hospital setting, as well as the overall prescribing pattern from 2006 to 2015, in the ACT and NSW area.
Materials and Methods
Study Design
This retrospective cross-sectional study analysed data from the Australian National University (ANU) Clinical Audit Project (CAP) database with ethics approval from the ethics committees from the Australian National University, NSW Health, ACT Health and Central Australian Aboriginal Congress (Northern Territory).
Data Collection
A standardised survey form [Appendix-1] was used to collect data on patient encounters in a range of clinical settings, including hospital wards, operating theatres, outpatient clinics, community general practice, and private rooms in the ACT, NT and NSW. Written consent was obtained from each patient prior to interview and data collection by students. Data was collected at consecutive patient encounters during clinical placement, over a set 2-week period every year from 2006-2012 and from 2014-2015. No data was collected during 2013 due to lapse in the ethics approval. To maintain confidentiality, all survey data was de-identified before entry into the ANU CAP database using standardised fields and International Classification of Primary Care, version 2 PLUS (ICPC-2+) diagnostic coding [Appendix-2] to ensure consistency and enable comparison with other data sets [8]. Antibiotics were grouped according to the Anatomical Therapeutic Chemical (ATC) classification system subgroup J01 ‘Antibiotics for systemic use’ [9].
Definition
In accordance with the ICD-10 definition, an URTI was defined as a disease caused by an infectious process affecting the upper respiratory tract (trachea, larynx, pharynx, paranasal sinuses or nose) [10].
Inclusion Criteria
Consequently, an encounter was included in the study if the diagnosis at the end of the encounter was defined as any of the following: rhinitis, rhinosinusitis, sinusitis, nasopharyngitis, tonsillitis, epiglottitis, otitis media, laryngitis, laryngeotracheitis, tracheitis or pharyngitis. These were identified in the database using the ICPC-2+ classifications [8]. Encounters were also included if the symptomology was consistent with an URTI – e.g., sneezing, sore throat, stuffy nose, coughing, chills, headaches, mucopurulent nasal discharge, or facial pain.
Statistical Analysis
Data was analysed using SPSS Version 22.0 Descriptive statistics (e.g., means, frequencies, percentages) were used to summarise patient demographic data. Chi square and t-tests were performed where appropriate. The correlation, direction, strength and significance of antibiotic use over the study duration were considered by fitting a simple regression line to the data. No data transformation was attempted. A p-value < 0.05 was considered to be significant.
Results
Characteristics of Patients Presenting with URTI
The total number of patient encounters recorded during the study period was 12,468. The proportion of patients presenting with URTIs was 5.6% (n=698) and their demographics are described in [Table/Fig-1].
Demographics of patients and prevalence of antibiotics prescription presenting with upper respiratory tract infections.
| Characteristic | Number (%)(n=698) | Antibioticprescribed n(%) | p-value |
|---|
| Age at presentation | Paediatric (<5 years) | 206 (29.5) | 72 (35.0) | 0.086 |
| Paediatric (6-17 years) | 86 (12.3) | 33 (38.4) |
| Adult (18-64 years) | 296 (42.4) | 136 (45.9) |
| Adult (64+ years) | 110 (15.8) | 48 (43.6) |
| Gender | Female | 375 (53.7) | 136 (42.1) | 0.727 |
| Male | 323 (46.3) | 153 (40.8) |
| Site | Hospital | 162 (23.2) | 52 (32.1) | 0.006* |
| Community | 536 (76.8) | 237 (44.2) |
| Rurality | Urban | 541 (77.5) | 218 (40.3) | 0.270 |
| Rural | 157 (22.5) | 71 (45.2) |
| Indigenous status | Aboriginal and/or TorresStrait Islander | 40 (5.7) | 11 (27.5) | 0.066 |
| Neither | 658 (94.3) | 278 (42.2) |
| Clinician gender | Male | 369 (66.2) | 164 (44.4) | 0.252 |
| Female | 188 (33.8) | 74 (39.4) |
| Missing | 141 (20.2) | |
| Presentation group | Localised (tonsillitis, sinusitis, otitis media)General (common cold) | | 109 (64.9)122 (33.7) | <0.001* |
| Presentation subgroup | TonsillitisSinusitisOtitis mediaCommon cold | | 25 (55.6)54 (71.1)44 (66.7)122 (33.7) | <0.001*2 |
* Data are presented as n (%). p-value is for antibiotics prescribed and is obtained by Pearson chi square with Yates correction.
1 Total of 141 were missing.
2 p-value remains the same when individual group (tonsillitis, sinusitis, and otitis media) were compared with common cold.
Comparison of URTIs Prescribed Antibiotics vs. Non-Antibiotic Interventions
Overall, the proportion of patients presenting with URTI prescribed an antibiotic was 42.7% (n=289) compared to non-antibiotic interventions (57.3%; n=409). This group was then stratified further as shown in [Table/Fig-1].
There was a significant difference in antibiotic prescribing for URTIs based on location, with fewer antibiotics prescribed in community GP (32.1%; n=52) compared to hospital (44.2%; n=237; p<0.05) [Table/Fig-1].
Antibiotics were more likely to be prescribed for URTI with localised symptoms (65.9%; n=109) compared to generalised symptoms (33.7%; n=122; p<0.01) [Table/Fig-1].
There was no significant difference for URTIs treated with antibiotics compared to non-antibiotic interventions based on patient age, patient gender, clinician gender, rurality (urban vs. rural) or Indigenous status.
Type of Antibiotic Prescribed for Urtis
Overall, the most frequently prescribed antibiotic class for URTI was penicillin (67.8%; n=196) followed by macrolides (17.0%; n=52), other beta-lactams (11.8%; n=34), and other antibiotics (6.57%; n=19).
Trends in URTI Antibiotic Prescribing
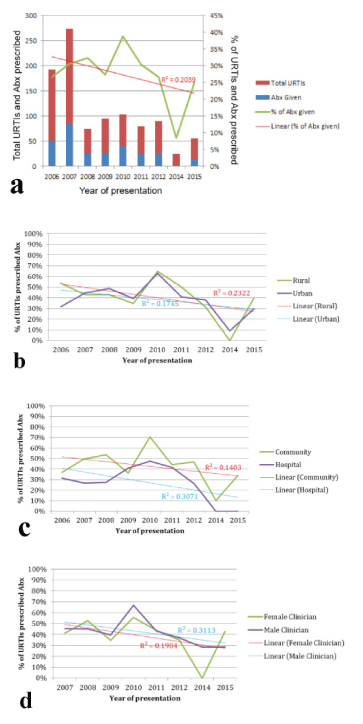
Over the course of the study period, 2006-2015, a decreasing trend was evident in the proportion of URTIs prescribed antibiotics (R2=0.204) [Appendix-3]. A similar trend was observed for antibiotic prescribing with respect to urban (R2=0.2322) and rural localities (R2=0.1745), male clinician gender (R2=0.313), community (R2= 0.1403) and for hospital (R2= 0.3071) and female clinician gender (R2=0.1984) [Table/Fig-2a-d].
Linear aggression analysis for trend of Upper Respiratory Tract Infections (URTIs) presentations and antibiotics (Abx) prescription by year of presentation for a) overall sample; b) patient location (rural/urban; c) healthcare facility setting (community/hospital; d) and clinicians gender (Male/Female) .
Discussion
Comparing Antibiotic Prescribing Practices
The factors identified as being significantly associated with antibiotic prescribing for URTIs in ACT and southeast NSW, were patient presentations in hospitals (cf. community practices) and the presence of localised URTI symptoms. Factors that had no significant association in this study included patient gender, patient age, indigenous status, practice rurality, or clinician gender.
The finding that hospital patients were more likely to be prescribed antibiotics reflects the relative increased severity of illness and the greater vulnerability of in-patients with infection compared to patients in the community. In terms of rurality of patient encounter, this study did not agree with other, albeit older, studies [6], which described higher rates of antibiotic use in rural locations. This may reflect recent improvements in the availability of medical facilities in rural Australia [11], where clinicians may be more confident in prescribing symptomatic relief under the assumption that they can seek further support if the patient’s condition deteriorates.
The higher rates of prescription antibiotics for URTIs with focalised symptomatology such as otitis media, tonsillitis and sinusitis (cf. generalised malaise, sore throat, rhinorrhoea and nasal congestion, etc., of the ‘common cold’) reflects improved compliance with the Antibiotic Therapeutic Guidelines, which directs antimicrobial therapy for these specific conditions but not for viral colds [12]. This improved compliance, in turn, may be a consequence of the effectiveness of antibiotic stewardship clinical standards, NPS education programs, and changes in attitudes of medical professionals to antibiotic prescribing.
The study’s results did not support the hypothesis of increased antibiotic prescribing in the extremes of age. There was no difference in prescribing patterns between age groups: <5 years, 6-17 years, 18-64 years and 85+ years (p=0.086). The effect of higher prescribing anticipated in the more vulnerable patient groups at the extremes of age may be masked by increased prescribing in the adult and general paediatric population (i.e., 6-64-year-old) due to the perceived or outright demand by patients and parents for antibiotics - which has been previously demonstrated to have a significant influence on antibiotic prescribing despite negative examination findings [13].
In previous studies, male clinicians were more likely to prescribe antibiotics to children under five for respiratory infections than female clinicians [14]. However, in this study, clinician gender was not significantly associated with prescribing patterns. This could reflect a multitude of factors such as increasing numbers of female doctors, changing attitudes to antibiotics in medical curricula, and retirement of older generations of doctors, who have previously been found more likely to prescribe medications than provide patient education, counselling or guidance [14].
Notably, in this study, there was no significant difference in antibiotic prescribing for indigenous and non-indigenous patients despite the Antibiotic Therapeutic Guidelines recommending routine use of antibiotics for indigenous patients with URTIs, due to a higher risk of developing non-suppurative complications [12]. This discrepancy may reflect that the majority of data collection occurred in urban private practice, rather than rural and remote communities and relied on patient identification as indigenous, which may be under-reported in interview-based survey (cf. self-report). Additionally, there were only a small number of Indigenous patients (n=40) in the study, which may affect the representativeness of the sample.
Prescribing Trends: 2006-2015
The Bettering the Evaluation and Care of Health (BEACH) study reported that 32% of patients presenting with URTI received clinical treatment (ranging from general and specific advice, counselling or education, to administrative processes); however, 59.2% of these patients did not receive medications [15].
This study showed that both the rate of URTI presentations to health services and rate of antibiotic prescription have decreased over the 2006-2015 study period. While the decrease in URTI presentations is not consistent with the BEACH study [15], the decline in antibiotic prescribing suggests that the aims of antibiotic stewardship standards are being achieved in the ACT and southeast NSW through programs such as NPS clinical and patient education campaigns.
Despite the overall downward trend, there is a notable increase in antibiotic prescribing in 2010 [Table/Fig-2]. This isolated deviation may be related to notably negative Australian media coverage of the 2009 H1N1 influenza pandemic [16], and predication of a second, potentially worse, wave in 2010 [17]. The resulting increased public perception of risk was associated with increased vigilance of URTI symptoms and greater consumer demand for antibiotics [16,18].
Limitation
The study may be limited by detection bias as a consequence of data collection occurring annually during the winter months. Moreover, given that the annual flu season varies year-to-year, interpretation of trends over the study period should take into consideration the timing of each year’s ‘flu season’ relative to data collection.
Selection bias should also be assumed, as the data collection was only carried out at select and limited sites where third-year ANU medical students were routinely assigned on placement. This restricted patient selection to predominantly smaller private practices in urban communities of ACT and southeast NSW. Thus, it could be anticipated that the findings are not representative of patients who access public health services (e.g., walk-in clinics) or bulk-billing centres.
Clinician gender data was missing from the database in 2006. Consequently, 2006 was excluded from this analysis, and a smaller sample was used to examine the influence of clinician gender on antibiotic prescribing.
The study is also limited by the lack of data on patient comorbidities, which may have strongly influenced clinician prescribing.
Conclusion
This study identified an overall downward trend in antibiotic prescribing rates in patients presenting with URTIs over the period between 2006-2015. This may suggest that antibiotic stewardship programs are making a positive impact on clinician prescribing behaviour. Nonetheless, continued concerted efforts are required to further improve responsible and appropriate antimicrobial use. Areas for particular focus in future stewardship campaigns may include improving clinician resistance to patient demand for antibiotics, and increasing confidence in prescribing for special populations such as indigenous peoples and the extremes of age.
Contributors’ Statement: All authors contributed to the conception and the study; Justin Friedman and Harrison Pickup undertook the statistical analyses; all authors drafted part of the manuscript. Hannah Glenn, Alexander A. Borecki and MEA led the writing of this paper. All authors contributed to the final manuscript and approved the submitted manuscript. This manuscript was a group project for final year medical students. Eight students were allocated to this project under supervision of a senior /lead supervisor.
Data sharing statement: All authors had full access to all of the data (including statistical reports and tables) in the study and can take responsibility for the integrity of the data and the accuracy of the data analysis.